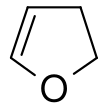
2,3-Dihydrofuran
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2,3-Dihydrofuran | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.013.407 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H6O | |||
| Molar mass | 70.091 g·mol−1 | ||
| Density | 0,927 g/mL | ||
| Boiling point | 54.6 °C (130.3 °F; 327.8 K)[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
2,3-Dihydrofuran is a heterocyclic compound. It is one of the simplest enol ethers and a position isomer of 2,5-dihydrofuran. It is a colorless volatile liquid.
It undergoes lithiation upon treatment with butyl lithium.[2]
References[]
- ^ Wilson, Christopher L. (December 1947). "Reactions of Furan Compounds. VII. Thermal Interconversion of 2,3-Dihydrofuran and Cyclopropane Aldehyde". Journal of the American Chemical Society. 69 (12): 3002–3004. doi:10.1021/ja01204a020.
- ^ Krzysztof Jarowicki; Philip J. Kocienski; Liu Qun (2002). "1,2-Metallate Rearrangement: (Z)-4-(2-Propenyl)-3-octen-1-ol". Org. Synth. 79: 11. doi:10.15227/orgsyn.079.0011.
Categories:
- Dihydrofurans
- Heterocyclic compound stubs