Acridone alkaloids

Acridone, parent compound of the acridone alkaloids.

Wine rue (Ruta graveolens)
Acridone alkaloids are natural products derived from acridone.[1]
Occurrence[]
Acridone alkaloids are found in bark, wood, leaves and roots of rue plants,[1] especially in roots and suspension cultures of rue.[2]
Examples[]
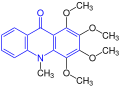
This group is named after the acridone. Further members are acronycin, melicopicin and rutacridone, among others:[1][3]
Acronycin
Melicopicin
Rutacridon
Properties[]
Many acridone alkaloids are methylated on the nitrogen atom and also have two oxygen functional groups, which can be free, alkylated or incorporated into rings. Acridone alkaloids show a blue-green fluorescence so that they can be detected with UV light. Some alkaloids of this group are effective against malaria pathogens. Furthermore, acronycin inhibits cell division.[2]
References[]
- ^ a b c Entry on Acridone. at: Römpp Online. Georg Thieme Verlag, retrieved 21 April 2020.
- ^ a b R. Hänsel, O.Sticher (2007), Pharmakognosie Phytopharmazie (in German) (8 ed.), Heidelberg: Springer Medizin Verlag, pp. 101,1326, ISBN 9783540265085
- ^ "Acridone alkaloid biosynthesis". Retrieved 22 April 2020.
Categories:
- Alkaloids by chemical classification