Arylide yellow
| Arylide yellow | |
|---|---|
| Hex triplet | #E9D66B |
| sRGBB (r, g, b) | (233, 214, 107) |
| HSV (h, s, v) | (51°, 54%, 91%) |
| CIELChuv (L, C, h) | (85, 71, 76°) |
| Source | [1] [2] |
| ISCC–NBS descriptor | Brilliant greenish yellow |
| B: Normalized to [0–255] (byte) | |
Arylide yellow, also known as Hansa yellow and monoazo yellow, is a family of organic compounds used as pigments. They are primarily used as industrial colorants including plastics, building paints and inks. They are also used in artistic oil paints, acrylics and watercolors. These pigments are usually semi-transparent and range from orange-yellow to yellow-greens. Related organic pigments are the diarylide pigments. Overall, these pigments have partially displaced the toxic cadmium yellow in the marketplace. Painters such as Alexander Calder and Jackson Pollock are known to have employed arylide yellow in their artworks.[1]
Production[]
The compound is obtained by azo coupling of aniline and acetoacetanilide or their derivatives. The class of compounds was discovered in Germany in 1909.[2]
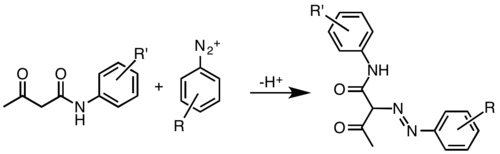 Synthesis of Hansa Yellow Pigments, R and R' represent a variety of substituents.
Synthesis of Hansa Yellow Pigments, R and R' represent a variety of substituents.
Examples[]
Members of this class include:
- 6 (CAS# 2512-29-0), derived from 4-chloro-2-nitroaniline (diazonium precursor) and aniline (acetoacetanilide precursor) to produce medium yellows.
- (CAS# 6486-23-3), derived from 4-chloro-2-nitroaniline (diazonium precursor) and (acetoacetanilide precursor) to produce greenish-yellows.
- Pigment Yellow 16 derived from 2,4-dichloroaniline (diazonium precursor) and tolidine (acetoacetanilide precursor) to produce medium-yellows.
- (CAS# 6358-31-2), derived from 2-methoxy-4-nitroaniline (diazonium precursor) and 2-methoxyaniline (acetoacetanilide precursor) to produce greenish-yellows.
See also[]
- List of colors
References[]
- ^ Arylide Yellow, Colourlex
- ^ K. Hunger. W. Herbst "Pigments, Organic" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2012. doi:10.1002/14356007.a20_371
- Shades of yellow
- Pigments
- Organic pigments
- Azo dyes
