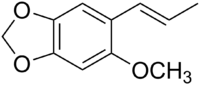
Carpacin
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
5-Methoxy-6-[(1E)-prop-1-en-1-yl]-2H-1,3-benzodioxole | |
| Other names
(E)-5-Methoxy-6-(prop-1-en-1-yl)benzo[d][1,3]dioxole
(E)-2-Methoxy-4,5-methylenedioxypropenylbenzene | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C11H12O3 | |
| Molar mass | 192.214 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Carpacin is a naturally occurring organic compound first isolated from the Carpano tree (an unidentified Cinnamomum species of the family Lauraceae which is native to Bougainville Island), from which it derives its name. It is also found in essential oils of the genus Crowea.[1]
Carpacin is a biosynthetic precursor of the more complex lignan-dimer, carpanone.[2] It is classified as a phenylpropanoid.
Carpacin has been prepared synthetically from sesamol[3] and has been studied for potential use as an insecticide[4] and inhibitor of carcinogenesis.[3]
References[]
- ^ Brophy, Joseph J.; Goldsack, Robert J.; Punruckvong, Acharaporn; Forster, Paul I.; Fookes, Christopher J.R. (1997). "Essential oils of the genus Crowea (Rutaceae)". Journal of Essential Oil Research. 9 (4): 401–409. doi:10.1080/10412905.1997.9700740.
- ^ J. Mohandas; M. Slaytor; T.R. Watson (1969). "Trans-2-methoxy-4,5-methylenedioxypropenylbenzene (carpacin) from a Cinnamomum sp. from Bougainville". Aust. J. Chem. 22 (8): 1803–1804. doi:10.1071/CH9691803.
- ^ a b Tsui-Hwa Tseng; Yen-Min Tsheng; Yean-Jang Lee; Hsing-Ling Hsu (2000). "Total Synthesis of Carpacin and Its Geometric Isomer as a Cancer Chemopreventer" (PDF). Journal of the Chinese Chemical Society. 47 (5): 1165–1169. doi:10.1002/jccs.200000157. Archived from the original (PDF) on 2005-09-14.
- ^ BH Alexander; SI Gertler; RT Brown; TA Oda; M Beroza (1959). "Synthesis of Methylenedioxyphenyl Compounds from Isosafrole and Sesamol". J. Org. Chem. 24 (10): 1504. doi:10.1021/jo01092a029.
Categories:
- Phenylpropenes
- O-Methylated phenylpropanoids
- Benzodioxoles