Cyclopentane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cyclopentane | |||
| Other names
pentamethylene
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.470 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| Properties | |||
| C5H10 | |||
| Molar mass | 70.1 g/mol | ||
| Appearance | clear, colorless liquid | ||
| Odor | mild, sweet | ||
| Density | 0.751 g/cm3 | ||
| Melting point | −93.9 °C (−137.0 °F; 179.2 K) | ||
| Boiling point | 49.2 °C (120.6 °F; 322.3 K) | ||
| 156 mg·l−1 (25 °C)[1] | |||
| Solubility | soluble in ethanol, acetone, ether | ||
| Vapor pressure | 45 kPa (20 °C) [2] | ||
| Acidity (pKa) | ~45 | ||
| -59.18·10−6 cm3/mol | |||
Refractive index (nD)
|
1.4065 | ||
| Hazards | |||
| Main hazards | Flammable[3] | ||
| NFPA 704 (fire diamond) | 
1
3
0 | ||
| Flash point | −37.2 °C (−35.0 °F; 236.0 K) | ||
| 361 °C (682 °F; 634 K) | |||
| Explosive limits | 1.1%-8.7%[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none[3] | ||
REL (Recommended)
|
TWA 600 ppm (1720 mg/m3)[3] | ||
IDLH (Immediate danger)
|
N.D.[3] | ||
| Related compounds | |||
Related compounds
|
cyclopropane, cyclobutane, cyclohexane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Cyclopentane (also called C pentane) is a highly flammable alicyclic hydrocarbon with chemical formula C5H10 and CAS number 287-92-3, consisting of a ring of five carbon atoms each bonded with two hydrogen atoms above and below the plane. It occurs as a colorless liquid with a petrol-like odor. Its melting point is −94 °C and its boiling point is 49 °C. Cyclopentane is in the class of cycloalkanes, being alkanes that have one or more rings of carbon atoms. It is formed by cracking cyclohexane in the presence of alumina at a high temperature and pressure.
It was first prepared in 1893 by the German chemist Johannes Wislicenus.[4]
Industrial usage[]
Cyclopentane is used in the manufacture of synthetic resins and rubber adhesives and also as a blowing agent in the manufacture of polyurethane insulating foam, and is found in many domestic appliances such as refrigerators and freezers as a refrigerant, replacing alternatives such as CFC-11 and HCFC-141b.[5]
Multiply alkylated cyclopentane (MAC) lubricants have low volatility and are used in some specialty applications.[6]
Formulation of cycloalkanes[]
Cycloalkanes can be formulated via a process known as catalytic reforming.
For example, 2-methylbutane can be reformed into cyclopentane, by use of a platinum catalyst. This is particularly well known in automobiles, as branched alkanes will burn much more readily.
Conformations[]

Envelope.
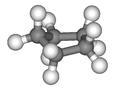
3D envelope.

Half-chair.
References[]
- ^ Record of cyclopentane in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 28 February 2015.
- ^ http://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0353
- ^ Jump up to: a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0171". National Institute for Occupational Safety and Health (NIOSH).
- ^ J. Wislicenus and W. Hentschel (1893) "Der Pentamethenylalkohol und seine Derivate" (Cyclopentanol and its derivatives), Annalen der Chemie, 275 : 322-330; see especially pages 327-330. Wislicenus prepared cyclopentane from cyclopentanone ("Ketopentamethen"), which is prepared by heating calcium adipate.
- ^ Greenpeace - Appliance Insulation Archived 2008-10-30 at the Wayback Machine
- ^ Pennzane - lubrication technology
External links[]
 Media related to Cyclopentane derivatives at Wikimedia Commons
Media related to Cyclopentane derivatives at Wikimedia Commons
- Cyclopentanes
- Hydrocarbon solvents
- Cycloalkanes




