From Wikipedia, the free encyclopedia
Fungisterol
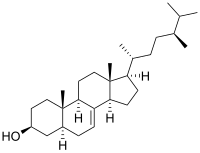
|
| Names
|
Preferred IUPAC name
(1R,3aR,5aS,7S,9aS,9bR,11aR)-1-[(2R,5S)-5,6-Dimethylheptan-2-yl]-9a,11a-dimethyl-2,3,3a,5,5a,6,7,8,9,9a,9b,10,11,11a-tetradecahydro-1H-cyclopenta[a]phenanthren-7-ol |
| Other names
5apha-ergost-7-en-3beta-ol
|
| Identifiers
|
|
|
|
3D model (JSmol)
|
|
| ChEBI
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.007.479 
|
| EC Number
|
|
|
|
|
| UNII
|
|
InChI=1S/C28H48O/c1-18(2)19(3)7-8-20(4)24-11-12-25-23-10-9-21-17-22(29)13-15-27(21,5)26(23)14-16-28(24,25)6/h10,18-22,24-26,29H,7-9,11-17H2,1-6H3/t19-,20+,21-,22-,24+,25-,26-,27-,28+/m0/s1 Key: PUGBZUWUTZUUCP-ZRKHGVCBSA-N
|
CC(C)[C@@H](C)CC[C@@H](C)[C@H]1CC[C@H]2C3=CC[C@H]4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@]12C
|
| Properties
|
|
|
C28H48O
|
| Molar mass
|
400.691 g·mol−1
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
| Infobox references
|
|
|
|
Chemical compound
Fungisterol is a bio-active sterol made by certain fungi.[1]
References[]
- ^ Kamilova, TA; Ekhvalova, TV (1989). "Resistance of yeasts to polyene antibiotics". Genetika. 25 (9): 1705–7. PMID 2689285.
Hidden categories:
- Articles without InChI source
- Articles without KEGG source
- Chembox CAS registry number linked
- ECHA InfoCard ID from Wikidata
- Pages using collapsible list with both background and text-align in titlestyle
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- All stub articles
