From Wikipedia, the free encyclopedia
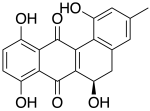
Landomycinone
|
| Names
|
| IUPAC name
(6R)-1,6,8,11-Tetrahydroxy-3-methyl-5,6-dihydro-7,12-tetraphenedione
|
| Identifiers
|
3D model (JSmol)
|
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
|
|
|
InChI=1S/C19H14O6/c1-7-4-8-6-12(23)16-17(13(8)11(22)5-7)19(25)15-10(21)3-2-9(20)14(15)18(16)24/h2-5,12,20-23H,6H2,1H3/t12-/m1/s1 Key: DMGKSWBAQZKXEJ-GFCCVEGCSA-N InChI=1/C19H14O6/c1-7-4-8-6-12(23)16-17(13(8)11(22)5-7)19(25)15-10(21)3-2-9(20)14(15)18(16)24/h2-5,12,20-23H,6H2,1H3/t12-/m1/s1 Key: DMGKSWBAQZKXEJ-GFCCVEGCBR
|
Cc1cc2c(c(c1)O)C3=C([C@@H](C2)O)C(=O)c4c(ccc(c4C3=O)O)O
|
| Properties
|
|
|
C19H14O6
|
| Molar mass
|
338.311
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
| Infobox references
|
|
|
|
Chemical compound
Landomycinone is the aglycone of the landomycins.[1]
See also[]
References[]
- ^ Bugaut, Xavier; Guinchard, Xavier; Roulland, Emmanuel (December 3, 2010). "Synthesis of the landomycinone skeleton". The Journal of Organic Chemistry. ACS Publications. 75 (23): 8190–8198. doi:10.1021/jo1018377. PMID 21028772.
Categories:
- Tetrols
- Angucyclines
- Organic compound stubs
Hidden categories:
- Orphaned articles from September 2013
- All orphaned articles
- Chemical articles without CAS registry number
- Articles without KEGG source
- Articles without UNII source
- Pages using collapsible list with both background and text-align in titlestyle
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- All stub articles
