Nucleocidin
| |
| Names | |
|---|---|
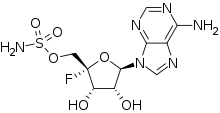
| Preferred IUPAC name
[(2S,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-2-fluoro-3,4-dihydroxyoxan-2-yl]methyl sulfamate | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H13FN6O6S | |
| Molar mass | 364.31 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Nucleocidin is a fluorine-containing nucleoside produced by Streptomyces calvus.[1]
During 1968 the attempts to identify nucleocidin were made and at that time it was assigned to a structure of a 9-adenyl-4' -sulfamoyloxypentofuranoside, which was mainly based on experiments from pmr and mass spectroscopy, as well as testing in chemical reactions. It was ultimately proven to have a structure of a 4' -fluoro-5' -O-sulphamoyladenosine.[2] Nucleocidin is an antibiotic that can be produced from Streptomyces calvus that grows on Indian land. It is essentially toxic for mammals, although nucleocidin is able to function against bacteria, such as gram negative, and gram positive. Moreover it can be used against trypanosomes.[3]
See also[]
References[]
- ^ Bartholomé, Axel; Janso, Jeffrey E; Reilly, Usa; O'Hagan, David (2017). "Fluorometabolite biosynthesis: isotopically labelled glycerol incorporations into the antibiotic nucleocidin in Streptomyces calvus". Organic & Biomolecular Chemistry. 15 (1): 61–64. doi:10.1039/c6ob02291j. hdl:10023/12031. PMID 27845468.
- ^ Shuman, Dennis A.; Robins, Morris J.; Robins, Roland K. (June 1970). "Synthesis of Nucleoside Sulfamates Related to Nucleocidin". Journal of the American Chemical Society. 92 (11): 3434–3440. doi:10.1021/ja00714a035. ISSN 0002-7863. PMID 5422764 – via JACS.
- ^ Carvalho, Maria F.; Oliveira, Rui S (October 2017). "Natural Production of Fluorinated Compounds and Biotechnological Prospects of the Fluorinate Enzyme". Critical Reviews in Biotechnology. 37 (7): 880–897. doi:10.1080/07388551.2016.1267109. hdl:10400.22/13870. ISSN 0738-8551. PMID 28049355. S2CID 27301409.
Categories:
- Nucleosides
- Organofluorides
- Fluorine-containing natural products
- Sulfamates
- Biochemistry stubs