Ortho-carborane

| |
| Names | |
|---|---|
| Other names
1,2-Dicarbadodecaborane(12), ortho-dicarbadodecaborane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.037.164 |
| EC Number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H12B10 | |
| Molar mass | 144.22 g·mol−1 |
| Appearance | colorless solid |
| Melting point | 320 °C (608 °F; 593 K) |
| Hazards | |
| GHS labelling: | |
 
| |
Signal word
|
Warning |
| H228, H302, H312, H332 | |
| P210, P240, P241, P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P312, P322, P330, P363, P370+P378, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ortho-carborane is the organoboron compound with the formula C2B10H12. The prefix ortho is derived from ortho. It is the most prominent carborane. This derivative has been considered for a wide range of applications from heat-resistant polymers to medical applications. It is a colorless solid that melts, without decomposition, at 320 °C.
Structure[]
The cluster has C2v symmetry.[1]
Preparation[]
Ortho-carborane is prepared by the addition of acetylenes to decaborane(14). Modern syntheses involve two stages, the first involving generation of an adduct of decaborane:[2][3]
- B10H14 + 2 SEt2 → B10H12(SEt2)2 + H2
In the second stage, the alkyne is installed as the source of two carbon vertices:[3]
- B10H12(SEt2)2 + C2H2 → C2B10H12 + 2 SEt2 + H2
Substituted acetylenes can be employed more conveniently than acetylene gas. For example bis(acetoxymethyl)acetylene adds to the decarborane readily.
- B10H12(SEt2)2 + C2(CH2O2CCH3)2 → C2B10H10(CH2O2CCH3)2 + 2 SEt2 + H2
The organic substituents are removed by ester hydrolysis followed by oxidation:[2]
- 3 C2B10H10(CH2O2CH3)2 + 10 KOH + + 8 KMnO4 → 3 C2B10H12 + 6 CH3CO2K + 8 MnO2 + 6 K2CO3 + 8 H2O
Reactions[]
Thermal rearrangement[]
Upon heating to 420 °C, it rearranges to form the meta isomer. The para isomer is produced by heating to temperatures above 600 °C.

Reduction and "reverse isomerization"[]
ortho-Carborane undergoes 2e- reduction when treated with a solution of lithium in ammonia. The result is the nido cluster 7,9-[C2B10H12]2-. In the dianion, the carbon vertices are not adjacent. The same cluster is produced by reduction of meta-carborane. Oxidation of the resulting 7,9-[C2B10H12]2- gives ortho-carborane.[4]
Deprotonation[]
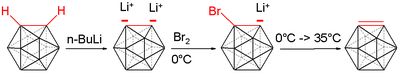
Treatment with organolithium reagents gives the dilithio derivative.[5]
- C2B10H12 + 2 BuLi → Li2C2B10H10 + 2 BuH
This dilithiated compound reacts with a variety of electrophiles, e.g. chlorophosphines, chlorosilanes, and sulfur.[6]
Base-degradation to dicarbollide[]
Base degradation of ortho carborane gives the anionic 11-vertex derivative, precursor to dicarbollide complexes:[7]
- C2B10H12 + NaOEt + 2 EtOH → Na+C2B9H12− + H2 + B(OEt)3
- Na+C2B9H12− + NaH → Na2C2B8H11 + H2
Dicarbollides (C2B8H112-) function ass ligands for transition metals and f-elements.[8] The dianion forms sandwich compounds, bis(dicarbollides). Dicarbollides, being strong electron donors, stabilize higher oxidation states, e.g. Ni(IV).
Deprotonation of carborane[]
The CH vertices of closo-dicarbadodecaboranes undergo deprotonation upon treatment with [organolithium]] reagents:[9]
- C2B10H12 + 2 BuLi → Li2C2B10H10 + 2 BuH
These dilithiated compounds react with a variety of electrophiles, e.g. chlorophosphines, chlorosilanes, and sulfur.[10] Many of the same compounds can be produced by hydroboration of alkynes:
- Li2C2B10H10 + 2 RX → R2C2B10H10 + 2 LiX
- L2B10H10 + RC2R → R2C2B10H10 + 2 L (L = MeCN, etc.)
ortho-Carborane can be converted to highly reactive carborynes with the formula B10C2H10.
History[]
The preparation of closo-dicarbadodecaboranes was reported independently by groups at Olin Corporation and the Reaction Motors Division of Thiokol Chemical Corporation working under the U.S. Air Force and published in 1963. These groups demonstrated the high stability in air of 1,2-closo-dodecaborane and related compounds, presented a general synthesis, described the transformation of substituents without destroying the carborane cluster, and demonstrated the ortho to meta isomerization.[11]
See also[]
References[]
- ^ Davidson, M. G.; Hibbert, T. G.; Howard, J. A. K.; Mackinnon, A.; Wade, K. (1996). "Definitive crystal structures of ortho-, meta- and para-carboranes: supramolecular structures directed solely by C–H⋯O hydrogen bonding to hmpa (hmpa = hexamethylphosphoramide)". Chem. Commun. (19): 2285–2286. doi:10.1039/CC9960002285.CS1 maint: uses authors parameter (link)
- ^ a b Charles R. Kutal David A. Owen Lee J. Todd (1968). closo‐1,2‐Dicarbadodecaborane(12). Inorganic Syntheses. 11. pp. 19–24. doi:10.1002/9780470132425.ch5.CS1 maint: uses authors parameter (link)
- ^ a b M. Frederick Hawthorne, Timothy D. Andrews, Philip M. Garrett, Fred P. Olsen, Marten Reintjes, Fred N. Tebbe, Les F. Warren, Patrick A. Wegner, Donald C. Young (1967). "Icosahedral Carboranes and Intermediates Leading to the Preparation of Carbametallic Boron Hydride Derivatives". Inorganic Syntheses. Inorganic Syntheses. 10. pp. 91–118. doi:10.1002/9780470132418.ch17. ISBN 9780470132418.CS1 maint: uses authors parameter (link)
- ^ Russell N. Grimes (2016). "10. Icosahedral Carboranes: 1,7-C2B10H12 and 1,12-C2B10H12". Carboranes, 3rd Edition. Elsevier. ISBN 9780128019054.
- ^ Popescu, A.-R.; Musteti, A. D.; Ferrer-Ugalde, A.; Viñas, C.; Núñez, R.; Teixidor, F. (2012). "Influential Role of Ethereal Solvent on Organolithium Compounds: The Case of Carboranyllithium". Chemistry – A European Journal. 18 (11): 3174–3184. doi:10.1002/chem.201102626. PMID 22334417.CS1 maint: uses authors parameter (link)
- ^ Jin, G.-X. (2004). "Advances in the Chemistry of Organometallic Complexes with 1,2-Dichalcogenolato-o-Carborane Ligands". Coord. Chem. Rev. 248 (7–8): 587–602. doi:10.1016/j.ccr.2004.01.002.
- ^ Plešek, J.; Heřmánek, S.; Štíbr, B. (1983). "Potassium dodecahydro-7, 8-dicarba-nido -undecaborate(1-), k[7, 8-c2 b9 h12 ], intermediates, stock solution, and anhydrous salt". Potassium dodecahydro-7,8-dicarba-nido-undecaborate(1-), k[7,8-C2B9H12], intermediates, stock solution, and anhydrous salt. Inorganic Syntheses. 22. pp. 231–234. doi:10.1002/9780470132531.ch53. ISBN 9780470132531.
- ^ Hawthorne, M. F.; Young, D. C.; Wegner, P. A. (1965). "Carbametallic Boron Hydride Derivatives. I. Apparent Analogs of Ferrocene and Ferricinium Ion". Journal of the American Chemical Society. 87 (8): 1818–1819. doi:10.1021/ja01086a053.
- ^ Popescu, A.-R.; Musteti, A. D.; Ferrer-Ugalde, A.; Viñas, C.; Núñez, R.; Teixidor, F. (2012). "Influential Role of Ethereal Solvent on Organolithium Compounds: The Case of Carboranyllithium". Chemistry – A European Journal. 18: 3174–3184. doi:10.1002/chem.201102626.CS1 maint: uses authors parameter (link)
- ^ Jin, G.-X. (2004). "Advances in the Chemistry of Organometallic Complexes with 1,2-Dichalcogenolato-o-Carborane Ligands". Coord. Chem. Rev. 248: 587–602. doi:10.1016/j.ccr.2004.01.002.
- ^ Heying, T. L.; Ager, J. W.; Clark, S. L.; Mangold, D. J.; Goldstein, H. L.; Hillman, M.; Polak, R. J.; Szymanski, J. W. (1963). "A New Series of Organoboranes. I. Carboranes from the Reaction of Decaborane with Acetylenic Compounds". Inorganic Chemistry. 2 (6): 1089–1092. doi:10.1021/ic50010a002.
- Organoboron compounds
- Cluster chemistry