Oxazines
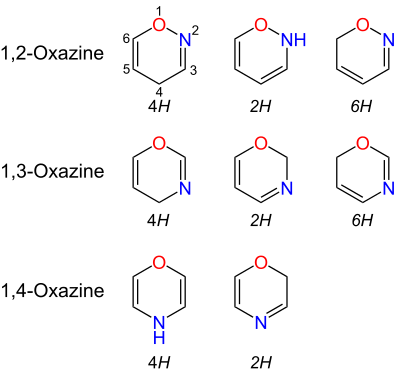
Oxazines are heterocyclic compounds containing one oxygen and one nitrogen atom in a doubly unsaturated six-membered ring. Isomers exist depending on the relative position of the heteroatoms and relative position of the double bonds.
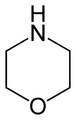
By extension, the derivatives are also referred to as oxazines; examples include ifosfamide and morpholine (tetrahydro-1,4-oxazine). A commercially available dihydro-1,3-oxazine is a reagent in the Meyers synthesis of aldehydes. Fluorescent dyes such as Nile red and Nile blue are based on the aromatic . Cinnabarine and cinnabaric acid are two naturally occurring dioxazines, being derived from biodegradation of tryptophan.[2]
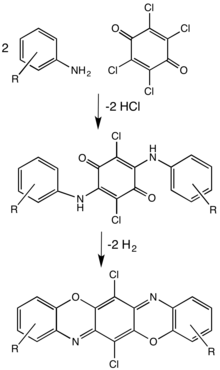
Dioxazines[]
Dioxazines are pentacyclic compounds consisting of two oxazine subunits. A commercially important example is the pigment pigment violet 23.[3]
Benzoxazines[]
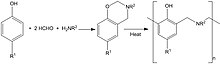
Benzoxazines are bicylic compounds formed by the ring fusion of a benzene ring with an oxazine. Polybenzoxazines are a class of polymers formed by the reaction of phenols, formaldehyde, and primary amines which on heating to ~200 °C (~400 °F) polymerise to produce polybenzoxazine networks.[5] The resulting high molecular weight thermoset polymer matrix composites are used where enhanced mechanical performance, flame and fire resistance compared to epoxy and phenolic resins is required.[6]
Images[]
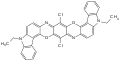
Pigment violet 23 is a commercially useful pigment.
References[]
- ^ Theophil Eicher, Siegfried Hauptmann, Andreas Speicher: The Chemistry of Heterocycles: Structures, Reactions, Synthesis, and Applications, 3rd, Completely Revised and Enlarged Edition, John Wiley & Sons, p. 442 (Oxazines, p. 442, at Google Books).
- ^ Stone, Trevor W.; Stoy, Nicholas; Darlington, L. Gail "An expanding range of targets for kynurenine metabolites of tryptophan" Trends in Pharmacological Sciences 2013, volume 34, pp. 136-143.
- ^ Chamberlain, Terence "Dioxazine violet pigments" from High Performance Pigments Edited by Smith, Hugh M. 2002,185-194. doi:10.1002/3527600493.ch12
- ^ Horst Tappe, Walter Helmling, Peter Mischke, Karl Rebsamen, Uwe Reiher, Werner Russ, Ludwig Schläfer and Petra Vermehren "Reactive Dyes"in Ullmann's Encyclopedia of Industrial Chemistry 2000, Wiley-VCH, Weinheim. doi:10.1002/14356007.a22_651
- ^ "BENZOXAZINE CHEMISTRY: A NEW MATERIAL TO MEET FIRE RETARDANT CHALLENGES OFAEROSPACE INTERIORS APPLICATIONS" (PDF).
- ^ Handbook of Benzoxazine Resins, ed. Hatsuo Ishida And Tarek Agag, Elsevier B.V., 2011, ISBN 978-0-444-53790-4
External links[]
- Oxazines at the US National Library of Medicine Medical Subject Headings (MeSH)
- Development of polymeric materials as a class of benzoxazines
- Oxazines