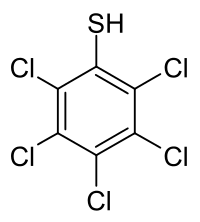
Pentachlorobenzenethiol
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,3,4,5,6-Pentachlorobenzene-1-thiol | |
| Other names
Pentachlorobenzenethiol, pentachlorothiophenol, PCBT
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.004.644 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6HCl5S | |
| Molar mass | 282.38 g·mol−1 |
| Appearance | Gray solid |
| Density | 1.7±0.1 g/cm3 |
| Melting point | 231.5 °C (448.7 °F; 504.6 K) |
| Insoluble | |
| Hazards | |
| GHS labelling: | |

| |
Signal word
|
Warning |
| Flash point | 144.6 °C (292.3 °F; 417.8 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Pentachlorobenzenethiol is a chemical compound from the group of thiols and organochlorine compounds.[1] The chemical formula is C
6HCl
5S.[2]
Synthesis[]
Pentachlorobenzenethiol can be obtained from hexachlorobenzene.[3]
Properties[]
Pentachlorobenzenethiol is a combustible gray solid with an unpleasant odor, practically insoluble in water. It has a monoclinic crystal structure.[4] The compound is not well-biodegradable and presumably bioaccumulable and toxic for aquatic organisms. Pentachlorobenzenethiol is itself a metabolite of hexachlorobenzene and is found in the urine and the excretions of animals receiving hexachlorobenzene.[5] Pentachlorobenzenethiol has a high potential for long-range transport via air as it is very slowly degraded in atmosphere.[6]
Applications[]
Pentachlorobenzenethiol is used in the rubber industry. The compound is added to rubber (both natural and synthetic) to facilitate processing (mastication).
See also[]
References[]
- ^ "Pentachlorobenzenethiol". chemindustry.com. Retrieved 9 June 2017.
- ^ "2,3,4,5,6-pentachlorobenzenethiol". Retrieved 9 June 2017.
- ^ Kulka, Marshall (1959). "Reaction of Hexachlorobenzene with Mercaptides". J. Org. Chem. 24 (4): 235–237. doi:10.1021/jo01084a021.
- ^ Wojcik, G.; Charbonneau, G. P.; Delugeard, Y.; Toupet, L. (1980). "The disordered crystal structure of pentachlorothiophenol". Acta Crystallogr. B36 (2): 506–507. doi:10.1107/S0567740880003706.
- ^ Koss, G.; Koransky, W.; Steinbach, K. (June 1976). "Studies on the toxicology of hexachlorobenzene: II. Identification and determination of metabolites". Archives of Toxicology. 35 (2): 107–114. doi:10.1007/BF00372764. PMID 947309.
- ^ "TC NES SUBGROUP ON IDENTIFICATION OF PBT AND VPVB SUBSTANCES". EChA. echa.europa.eu. Retrieved 9 June 2017.
External links[]
- Benzene derivatives
- Thiols
- Organochlorides
- Foul-smelling chemicals