Phosphoramidate
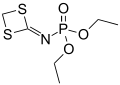
Phosphoramidates (sometimes also called amidophosphates) are a class of phosphorus compounds structurally related to phosphates (or organophosphates) via the substitution of an OR for a NR2. They are derivatives of phosphoramidic acids O=P(OH)(NR2)2, O=P(OH)2(NR2).
A phosphorodiamidate (or diamidophosphate) is a phosphate that has two of its OH groups substituted by NR2 groups to give a species with the general formula O=P(OH)(NH2)2. The substitution of all three OH groups gives the phosphoric triamides (O=P(NR2)3), which are commonly referred to as phosphoramides.[1]
Examples[]
Two examples of natural phosphoramidates are phosphocreatine and the phosphoramidate formed when histidine residues in histidine kinases are phosphorylated.[2] An example of a phosphorodiamidate is morpholino which is used in molecular biology.
See also[]
References[]
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "phosphoramides". doi:10.1351/goldbook.A00484
- ^ Stock, Ann M.; Robinson, Victoria L.; Goudreau, Paul N. (2000-07-01). "Two-Component Signal Transduction". Annual Review of Biochemistry. 69: 183–215. doi:10.1146/annurev.biochem.69.1.183. PMID 10966457.
- Phosphoramidates
- Functional groups