Stigmastane

| |
| Names | |
|---|---|
| IUPAC name
Stigmastane[1]
| |
| Preferred IUPAC name
(1R,3aS,3bR,5aΞ,9aS,9bS,11aR)-1-[(2R,5R)-5-Ethyl-6-methylheptan-2-yl]-9a,11a-dimethylhexadecahydro-1H-cyclopenta[a]phenanthrene | |
| Identifiers | |
3D model (JSmol)
|
|
| 8170826 | |
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C29H52 | |
| Molar mass | 400.735 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Stigmastane or 24R-ethylcholestane is a tetracyclic triterpene, along with cholestane and ergostane, this sterane is used as a biomarker for early eukaryotes.[2]

5α-Stigmastane
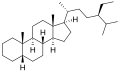
5β-Stigmastane
See also[]
- Stigmastanol (Stigmastan-3β-ol)
- β-Sitosterol (Stigmast-5-en-3β-ol)
- Stigmasterol (Stigmast-5,22-dien-3β-ol)
References[]
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 1531. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ Brocks, Jochen J.; Jarrett, Amber J. M.; Sirantoine, Eva; Hallmann, Christian; Hoshino, Yosuke; Liyanage, Tharika (2017). "The rise of algae in Cryogenian oceans and the emergence of animals". Nature. 548 (7669): 578–581. Bibcode:2017Natur.548..578B. doi:10.1038/nature23457. PMID 28813409. S2CID 205258987.
Categories:
- Triterpenes

