Cresol
| Isomers of Cresol[1][2][3][4] | ||||
|---|---|---|---|---|
| Skeletal formula | 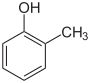 |
 |
||
| Ball-and-stick model | 
|

|
| |
| General | ||||
| Common name | o-cresol | m-cresol | p-cresol | |
| Preferred IUPAC name | 2-methylphenol | 3-methylphenol | 4-methylphenol | |
| Systematic name | 2-methylbenzenol | 3-methylbenzenol | 4-methylbenzenol | |
| Other names | ortho-Cresol 2-Hydroxytoluene |
meta-Cresol 3-Hydroxytoluene |
para-Cresol 4-Hydroxytoluene | |
| Molecular formula | C7H8O | |||
| SMILES | Oc1c(C)cccc1 | Oc1cc(C)ccc1 | Oc1ccc(C)cc1 | |
| Molar mass | 108.14 g/mol | |||
| Appearance at room temperature and pressure |
colorless crystals | thicker liquid | greasy-looking solid | |
| CAS number | [95-48-7] | [108-39-4] | [106-44-5] | |
| mixture of cresols (tricresol): [1319-77-3] | ||||
| Properties | ||||
| Density and phase | 1.05 g/cm3, solid | 1.03 g/cm3, liquid | 1.02 g/cm3, liquid | |
| Solubility in pure water at 20−25 °C |
2.5 g/100 ml | 2.4 g/100 ml | 1.9 g/100 ml | |
| soluble in strongly alkaline water | ||||
| Melting point | 29.8 °C (303.0 K) | 11.8 °C (285.0 K) | 35.5 °C (309.7 K) | |
| Boiling point | 191.0 °C (464.2 K) | 202.0 °C (475.2 K) | 201.9 °C (475.1 K) | |
| Acidity (pKa) | 10.287 | 10.09 | 10.26 | |
| Viscosity | solid at 25 °C | ? cP at 25 °C | solid at 25 °C | |
| Structure | ||||
| Dipole moment | 1.35 D | 1.61 D | 1.58 D | |
| Hazards | ||||
| SDS | ||||
| Main hazards | flammable, ingestion and inhalation toxicity hazard | |||
| Flash point | 81 °C c.c. | 86 °C | 86 °C c.c. | |
| R/S statement | R24/25-R34 ((S1/2)-)S36/37/S39-S45 | |||
| RTECS number | ||||
| Related compounds | ||||
| Related phenols | xylenols | |||
| Related compounds | bromo cresol | |||
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | ||||
Cresols (also hydroxytoluene) are a group of aromatic organic compounds. They are widely-occurring phenols (sometimes called phenolics) which may be either natural or manufactured. They are also categorized as methylphenols. Cresols commonly occur as either solids or liquids because their melting points are generally close to room temperature. Like other types of phenols, they are slowly oxidized by exposure to air, and the resulting impurities often give the samples a yellow to brownish red tint. Cresols have an odor characteristic to that of other simple phenols, reminiscent to some of a "coal tar" smell. The name "cresol" is an adduct of phenol and their traditional source, creosote.
Structure and production[]
In its chemical structure, a molecule of cresol has a methyl group substituted onto the ring of phenol. There are three forms (isomers) of cresol: ortho-cresol (o-cresol), meta-cresol (m-cresol), and para-cresol (p-cresol). These forms occur separately or as a mixture, which can also be called cresol or more specifically, tricresol.[citation needed] About half of the world's supply of cresols are extracted from coal tar. The rest is produced by hydrolysis of chlorotoluenes or the related sulfonates. Another method entails methylation of phenol with methanol over a solid acid catalyst, often comprising magnesium oxide or alumina. Temperatures above 300 °C are typical. Anisole converts to cresols under these conditions.[5][6]
Applications[]
Cresols are precursors or synthetic intermediates to other compounds and materials, including plastics, pesticides, pharmaceuticals, and dyes.[6]
For cresol bactericides or disinfectants the mechanism of action is due to the destruction of bacterial cell membranes.[7][8]
Most recently, cresols have been used to create a breakthrough in manufacturing carbon nanotubes at scale that are separated and not twisted, without additional chemicals that change the surface properties of the nanotubes.[9][10]
Commercial examples[]
Creolin, a 19th-century disinfectant.
Derivatives of p-cresol include:
- Butylated hydroxytoluene, a common antioxidant
Derivatives of o-cresol include:
- Indo-1, a popular calcium indicator
- MCPA, (4-chloro-2-methylphenoxy)acetic acid
- MCPB, 4-(4-chloro-2-methylphenoxy)butanoic acid
- Mecoprop, (RS)-2-(4-chloro-2-methylphenoxy)propanoic acid
- the amine atomoxetine, (3R)-N-methyl-3-(2-methylphenoxy)-3-phenylpropan-1-amine
- the diol mephenesin, 3-(2-methylphenoxy)propane-1,2-diol
Derivatives of m-cresol include:
- Amylmetacresol, an antiseptic
- Bevantolol, (RS)-[2-(3,4-dimethoxyphenyl)ethyl][2-hydroxy-3-(3-methylphenoxy)propyl]amine
- Bromocresol green
- Bupranolol, a non-selective beta blocker
- Chloro-m-cresol which is used as a household disinfectant
- Tolimidone, 5-(3-methylphenoxy)pyrimidin-2(1H)-one
Health effects[]
When cresols are inhaled, ingested, or applied to the skin, they can be very harmful. Effects observed in people include irritation and burning of skin, eyes, mouth, and throat; abdominal pain and vomiting; heart damage; anemia; liver and kidney damage; facial paralysis; coma; and death.
Breathing high levels of cresols for a short time results in irritation of the nose and throat. Aside from these effects, very little is known about the effects of breathing cresols, for example, at lower levels over longer times.
Ingesting high levels results in kidney problems, mouth and throat burns, abdominal pain, vomiting, and effects on the blood and nervous system.
Skin contact with high levels of cresols can burn the skin and damage the kidneys, liver, blood, brain, and lungs.
Short-term and long-term studies with animals have shown similar effects from exposure to cresols. No human or animal studies have shown harmful effects from cresols on reproduction.
It is not known what the effects are from long-term ingestion or skin contact with low levels of cresols.
The Occupational Safety and Health Administration has set a permissible exposure limit at 5 ppm (22 mg/m3) over an eight-hour time-weighted average, while the National Institute for Occupational Safety and Health recommends a limit of 2.3 ppm (10 mg/m3).[11]
See also[]
References[]
- ^ o-CRESOL (ICSC)
- ^ m-CRESOL (ICSC)
- ^ p-CRESOL (ICSC)
- ^ Pubchem. "o-cresol". pubchem.ncbi.nlm.nih.gov. Retrieved 2018-01-16.
- ^ W. W. Hartman (1923). "p-Cresol". Organic Syntheses. 3: 37. doi:10.15227/orgsyn.003.0037.
- ^ Jump up to: a b Helmut Fiegein "Cresols and Xylenols" in Ullmann's Encyclopedia of Industrial Chemistry" 2007; Wiley-VCH, Weinheim. doi:10.1002/14356007.a08_025
- ^ Judis, Joseph (1962). "Studies on the Mechanism of Action of Phenolic Disinfectants I". Journal of Pharmaceutical Sciences. 51 (3): 261–265. doi:10.1002/jps.2600510317. PMID 14452711.
- ^ "IDENTIFICATION Name Cresol". DrugBank Online. 12 June 2020.
- ^ "Making carbon nanotubes as usable as common plastics: Researchers discover that cresols disperse carbon nanotubes at unprecedentedly high concentrations". ScienceDaily, Northwestern University. 15 May 2018. Archived from the original on 2018-05-16.
- ^ Chiou, Kevin; Byun, Segi; Kim, Jaemyung; Huang, Jiaxing (29 May 2018). "Additive-free carbon nanotube dispersions, pastes, gels, and doughs in cresols". Proceedings of the National Academy of Sciences. 115 (22): 5703–5708. Bibcode:2018PNAS..115.5703C. doi:10.1073/pnas.1800298115. PMC 5984515. PMID 29760075.

- ^ Documentation for Immediately Dangerous To Life or Health Concentrations (IDLHs) - Cresol (o, m, p isomers)
- Antiseptics
- Hazardous air pollutants
- Cresols