Pegvaliase
 | |
| Clinical data | |
|---|---|
| Pronunciation | peg val' i ase |
| Trade names | Palynziq |
| Other names | Pegvaliase-pqpz; PEG-PAL; RAvPAL-PEG |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618057 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
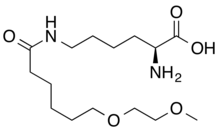
| Formula | C15H30N2O5 |
| Molar mass | 318.414 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Pegvaliase, sold under the brand name Palynziq, is a medication for the treatment of the genetic disease phenylketonuria.[2][3] Chemically, it is a pegylated derivative of the enzyme phenylalanine ammonia-lyase that metabolizes phenylalanine to reduce its blood levels.[4]
It was approved by the Food and Drug Administration for use in the United States in 2018.[2] The U.S. Food and Drug Administration (FDA) considers it to be a first-in-class medication.[5]
References[]
- ^ a b "Palynziq". Therapeutic Goods Administration (TGA). 23 July 2021. Retrieved 5 September 2021.
- ^ a b "FDA approves a new treatment for PKU, a rare and serious genetic disease" (Press release). Food and Drug Administration. May 24, 2018.
- ^ Mahan KC, Gandhi MA, Anand S (April 2019). "Pegvaliase: a novel treatment option for adults with phenylketonuria". Current Medical Research and Opinion. 35 (4): 647–651. doi:10.1080/03007995.2018.1528215. PMID 30247930.
- ^ "Palynziq". BioMarin Pharmaceutica.
- ^ New Drug Therapy Approvals 2018 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2019. Retrieved 16 September 2020.
External links[]
- "Pegvaliase". Drug Information Portal. U.S. National Library of Medicine.
Categories:
- Drugs acting on the gastrointestinal system and metabolism
- Recombinant proteins
- Orphan drugs
- Gastrointestinal system drug stubs