From Wikipedia, the free encyclopedia
Peonidin-3-O-glucoside
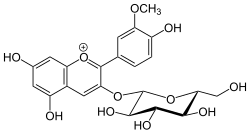
|
| Names
|
Preferred IUPAC name
5,7-Dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-3-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-1λ4-benzopyran-1-ylium |
| Other names
Peonidin-3-glucoside
Peonidin 3-O-glucoside
|
| Identifiers
|
|
|
|
3D model (JSmol)
|
|
| ChEBI
|
|
| ChemSpider
|
|
|
|
|
|
|
|
InChI=1S/C22H22O11/c1-30-15-4-9(2-3-12(15)25)21-16(7-11-13(26)5-10(24)6-14(11)31-21)32-22-20(29)19(28)18(27)17(8-23)33-22/h2-7,17-20,22-23,27-29H,8H2,1H3,(H2-,24,25,26)/p+1/t17-,18-,19+,20-,22-/m1/s1 Key: ZZWPMFROUHHAKY-OUUKCGNVSA-O
|
COC1=C(C=CC(=C1)C2=C(C=C3C(=CC(=CC3=[O+]2)O)O)OC4C(C(C(C(O4)CO)O)O)O)O
|
| Properties
|
|
|
C
22H
23O+
11
C22H23O11Cl (chloride)
|
| Molar mass
|
463.41 g/mol
498.9 g/mol (chloride)
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
| Infobox references
|
|
|
|
Chemical compound
Peonidin-3-O-glucoside is anthocyanin. It is found in fruits and berries, in red Vitis vinifera grapes and red wine,[1] in red onions and in purple corn.[2] It is dark red to purple in colour.
See also[]
- Phenolic compounds in wine
References[]
|
|---|
| 3-Hydroxyanthocyanidins | |
|---|
| 3-Deoxyanthocyanidins | |
|---|
| O-Methylated anthocyanidins | |
|---|
Anthocyanins
(anthocyaninidin glycosides) | Glucosides:
Diglucosides:
Others glycosides:
|
|---|
| Acylated anthocyanins | | Acetylated anthocyanins |
|
|---|
Coumaroylated anthocyanins
(cis- and trans-) | |
|---|
| Caffeoylated anthocyanins | |
|---|
| Malonylated anthocyanins |
- (malvidin 3-(6″-malonylglucoside)-5-glucoside)
|
|---|
| Acylated anthocyanin diglycosides |
- ()
- ()
- (petunidin 3-[6-O-(4-O-(E)-p-coumaroyl-O-α-l-rhamnopyranosyl)-β-D-glucopyranoside]-5-O-β-D-glucopyranoside)
- Violdelphin (Delphinidin 3-rutinoside-7-O-(6-O-(4-(6-O-(4-hydroxybenzoyl)-β-D-glucosyl)oxybenzoyl)-β-D-glucoside)
|
|---|
|
|---|
| Flavanol-anthocyanin adducts | |
|---|
| Miscellaneous | |
|---|
Categories:
- O-methylated anthocyanins
- Aromatic compound stubs
Hidden categories:
- Webarchive template wayback links
- Chemical articles with multiple compound IDs
- Multiple chemicals in an infobox that need indexing
- Chemical articles with multiple CAS registry numbers
- Articles without KEGG source
- Articles without UNII source
- Pages using collapsible list with both background and text-align in titlestyle
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- All stub articles
