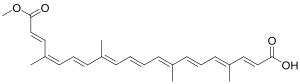
Bixin
| |

| |
| Names | |
|---|---|
| IUPAC name
(2E,4E,6E,8E,10E,12E,14E,16Z,18E)-20-Methoxy-4,8,13,17-tetramethyl-20-oxoicosa-2,4,6,8,10,12,14,16,18-nonaenoic acid
| |
| Other names
cis-Bixin; α-Bixin; 9-cis-6,6'-Diapo-ψ,ψ-carotenedioic acid, 6-methyl ester
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.027.499 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C25H30O4 | |
| Molar mass | 394.511 g·mol−1 |
| Appearance | Orange crystals |
| Insoluble | |
| Hazards | |
| NFPA 704 (fire diamond) | 
1
1
0 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bixin is an apocarotenoid found in the seeds of the achiote tree (Bixa orellana)[2] from which it derives its name. It is commonly extracted from the seeds to form annatto, a natural food coloring, containing about 5% pigments of which 70-80% are bixin.[3]
 red seeds of the achiote tree
red seeds of the achiote tree
Bixin is chemically unstable when isolated and converts via isomerization into trans-bixin (β-bixin), the double-bond isomer.[1]
 Chemical structure of trans-bixin
Chemical structure of trans-bixin
Bixin is soluble in fats and alcohols but insoluble in water. Upon exposure to alkali, the methyl ester is hydrolyzed to produce the dicarboxylic acid norbixin, a water-soluble derivative.
 Chemical structure of norbixin
Chemical structure of norbixin
References[]
- ^ a b Merck Index, 11th Edition, 1320
- ^ Bouvier, Florence; Dogbo, Odette; Camara, Bilal (2003). "Biosynthesis of the Food and Cosmetic Plant Pigment Bixin (Annatto)". Science. 300 (5628): 2089–2091. ISSN 0036-8075.
- ^ Executive Summary Bixin Archived July 21, 2011, at the Wayback Machine, National Toxicology Program
Categories:
- Apocarotenoids
- E-number additives
