Osteogenesis imperfecta
| Osteogenesis imperfecta (OI) | |
|---|---|
| Other names | Brittle bone disease,[1] Lobstein syndrome,[1]: 5 fragilitas ossium,[2] Vrolik disease,[1]: 5 osteopsathyrosis idiopathica[3]: 347 |
 | |
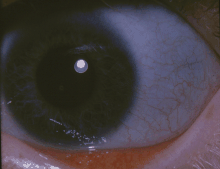
| Blue sclerae, as in the eyes of the girl above, are a classic non-pathognomonic sign of OI. | |
| Pronunciation | |
| Specialty | Pediatrics, medical genetics, orthopedics |
| Symptoms | Bones that break easily, blue tinge to the whites of the eye, short height, loose joints, hearing loss[2][6] |
| Onset | Birth[6] |
| Duration | Long term[6] |
| Causes | Genetic (autosomal dominant or de novo mutation)[2] |
| Diagnostic method | Based on symptoms, DNA testing[6] |
| Prevention | Pre-implantation genetic diagnosis |
| Management | Healthy lifestyle (exercise, no smoking), metal rods through the long bones[6] |
| Medication | Bisphosphonates[7] |
| Prognosis | Depends on the type[6] |
| Frequency | 1 in 10,000–20,000 people[2] |
Osteogenesis imperfecta (IPA: /ˌɒstioʊˈdʒɛnəsɪs ˌɪmpɜːrˈfɛktə/;[4][5] OI), also known as brittle bone disease, is a group of genetic disorders that mainly affect the bones.[2][1]: 85 It results in bones that break easily.[2] The range of symptoms may be mild to severe.[8]: 1512 Symptoms found in various types of OI include a blue tint to the whites of the eye (sclerae), short stature, loose joints, hearing loss, breathing problems[6] and problems with the teeth (dentinogenesis imperfecta).[8]
Potentially life-threatening complications, all of which become more common in more severe OI, include: tearing (dissection) of the major arteries, such as the aorta;[1]: 333 [9] pulmonary insufficiency secondary to distortion of the ribcage;[1]: 335–341 [10] and basilar invagination.[11]: 106–107
The underlying mechanism is usually a problem with connective tissue due to a lack of, or poorly formed, type I collagen.[8]: 1513 In more than 90% of cases, OI occurs due to mutations in the COL1A1 or COL1A2 genes.[2] These genetic problems may be inherited from a person's parents in an autosomal dominant manner, but may also occur via a new mutation—de novo.[2][12] There are four main types, with type I being the least severe and type II the most severe.[2] As of August 2021, 19 different genes are known to cause the 21 documented types of OI.[13][14] Diagnosis is often based on symptoms and may be confirmed by a collagen biopsy and/or a DNA test.[6]
Although there is no cure,[6] OI does not have a major effect on life expectancy,[1]: 461 [12] and many people with OI can achieve a significant agree of autonomy.[15] Maintaining a healthy lifestyle by exercising and avoiding smoking can help prevent fractures.[6] Treatment may include acute care of broken bones, pain medication, physical therapy, mobility aids such as leg braces or wheelchairs, and rodding surgery,[6] a type of surgery that puts metal intramedullary rods along the long bones (such as the femur) in an attempt to strengthen them.[6] Evidence also supports the use of medications of the bisphosphonate class, such as pamidronate, to increase bone density.[16] Bisphosphonates are especially effective in children,[17] however it is unclear if they lead to increases in quality of life or decrease the incidence of fractures.[7]
OI affects about one in 10,000 to 20,000 people.[2] Outcomes depend on the genetic cause of the disorder (its type), but death during childhood is rare.[6] Moderate to severe OI primarily affects mobility; if rodding surgery is performed during childhood, some of those with more severe types of OI may gain the ability to walk.[18] The condition has been described since ancient history.[19] The Latinate term osteogenesis imperfecta came into use in 1849 and literally translates to "imperfect bone formation".[19][20]: 683
Signs and symptoms[]
Orthopedic[]
The main symptom of OI is fragile, low mineral density bones; all types of OI have some bone involvement.[8] In moderate and especially severe OI, the long bones may be bowed, sometimes extremely so.[21] The weakness of the bones causes them to fracture easily; a study in Pakistan found an average of 5.8 fractures per year in untreated children.[22] Fractures typically occur much less after puberty, but begin to increase again in women after menopause and in men between the ages of 60 and 80.[1]: 486
Joint hypermobility is also a common sign of OI, thought to be because the affected genes are the same as those that cause some types of Ehlers–Danlos syndrome.[8]: 1513 (One OI mutation also causes combined Ehler–Danlos syndrome: "OIEDS1".)[23][24]
Otologic[]
By the age of 50, about 50% of adults with OI experience significant hearing loss, much earlier as compared to the general population.[25] Hearing loss in OI may or may not be associated with visible deformities of the ossicles and inner ear.[26] Hearing loss frequently begins during the second, third, and fourth decades of life, and may be conductive, sensorineural, or mixed in nature.[27] If hearing loss does not occur by age 50, it is significantly less likely to occur in the years afterwards.[25]
Although rare, OI-related hearing loss can also begin in childhood; in a study of forty-five children aged four to sixteen, two were found to be affected, aged 11 and 15.[28]
Hearing loss is most common in type I OI; it is less common in types III and IV.[1]: 294–296 Other parts of the inner ear may also be affected by OI. causing balance issues; however, only small studies have found links between vertigo and OI.[1]: 308
Neurological[]
OI is associated with a number of neurological abnormalities, usually involving the central nervous system, due to deformities in the skeletal structures surrounding it. Neurological complications, especially basilar invagination caused by upwards migration of the odontoid process,[29][11]: 106–107 may adversely affect life expectancy; neurosurgery may be needed to correct severe abnormalities when they risk the patient's life or cause intolerable neurological deficits.[29][11]: 106–107
Gastrointestinal[]
OI, especially its severe form type III, may be associated with recurrent abdominal pain and chronic constipation, according to two studies on patients affected by OI.[30][31] Chronic constipation is thought to be most common in patients with asymmetric pelvises (acetabular protrusion).[1]: 377 [31]
Cardiopulmonary[]
Type I collagen is present all throughout the circulatory and respiratory systems: from the ventricles of the heart itself, to the heart valves, to the vasculature,[1]: 329 and as an integral part of the connective tissue of the lungs.[1]: 336 As such, cardiovascular complications, among them aortic insufficiency, aortic aneurysm, and arterial dissections, are sometimes comorbid with OI,[1]: 333 but not as often comorbid as with Marfan syndrome.[1]: 332
Respiratory illnesses are a major cause of death in OI.[10][1]: 335 The most obvious source of respiratory problems in OI is pulmonary insufficiency caused by problems in the architecture of the thoracic wall.[1]: 341 However, respiratory tract infections, such as pneumonia, are also more fatal among those with OI than the general population.[10][32] Those with more severe ribcage deformities were found to have worse lung restriction in a small-scale 2012 study involving 22 Italian patients with OI types III and IV, plus 26 non-affected controls.[10]
Classification[]
There are two typing systems for OI in modern use. The first, created by David Sillence in 1979, classifies patients into four types, or syndromes, according to their clinical presentation, without taking into account the genetic cause of their disease.[33]: 114–115 [34] The second system expands on the Sillence model, but assigns new numbered types genetically as they are found.[35][34] Therefore, people with OI can be described as having both a clinical type and a genetic type, which may or may not be equivalent.[34]
Type I is the most common, and 90% of cases result from mutations to either COL1A1 or COL1A2.[2] Symptoms vary a lot between types, as well as vary from person to person, even in the same family.[36]
As of 2021, 21 genetic types of OI have been discovered:[13][14]
| Type | Description | Gene | OMIM | Mode of inheritance | Incidence |
|---|---|---|---|---|---|
| I | mild | Null COL1A1 allele | 166200 | autosomal dominant, 34% de novo[37] | 1 in 30,000[38] |
| II | severe and usually lethal in the perinatal period | COL1A1, COL1A2 | 166210 | autosomal dominant, ≈100% de novo[37] | 1 in 40,000[39] to 1 in 100,000[38] |
| III | considered progressive and deforming | COL1A1, COL1A2 | 259420 | autosomal dominant, 85% de novo[37] | 1 in 60,000[38] |
| IV | deforming, but with normal sclerae[1]: 294–296 | COL1A1, COL1A2 | 166220 | autosomal dominant, 50% de novo[37] | 1 in 30,000[1]: 21 |
| V | shares the same clinical features of IV, but has unique histologic findings ("mesh-like") | IFITM5 | 610967 | autosomal dominant[40][41] | Unknown
|
| VI | shares the same clinical features of IV, but has unique histologic findings ("fish scale") | SERPINF1 | 610968 | autosomal recessive[40] | |
| VII | clinically type III,[7] associated with cartilage associated protein | CRTAP | 610682 | autosomal recessive[40] | |
| VIII | clinically III,[7] associated with the protein leprecan | LEPRE1 | 610915 | autosomal recessive | |
| IX | normal collagen, but still causes disease clinically III[7] | PPIB | 259440 | autosomal recessive | |
| X | clinically III,[7] causes defects in the collagen chaperone protein HSP47 | SERPINH1 | 613848 | autosomal recessive | |
| XI | clinically III,[7] associated with the protein FKBP65 | FKBP10 | 610968 | autosomal recessive | |
| XII | clinically III,[7] causes hearing loss in most affected, related to a faulty osteoblast transcription factor | SP7 | 613849 | autosomal recessive | |
| XIII | clinically III,[7] related to faulty processing of collagen | BMP1 | 614856 | autosomal recessive | |
| XIV | of variable severity | TMEM38B | 615066 | autosomal recessive | |
| XV | clinically III, disrupts anabolic signaling of osteoblast cells | WNT1 | 615220 | autosomal recessive | |
| XVI | severe, sometimes lethal; causes missing transcription factor | CREB3L1 | 616229 | autosomal recessive | |
| XVII | severe, causes defective glycoprotein which binds to collagen | SPARC | 616507 | autosomal recessive | |
| XVIII | moderate, causes defects in FAM46A protein | TENT5A | 617952 | autosomal recessive | |
| XIX | moderate/severe, causes defective regulated intramembrane proteolysis | MBTPS2 | 301014 | X-linked recessive | |
| XX | clinically III, disrupts Wnt signaling[42] | MESD | 618644 | autosomal recessive | |
| XXI | causes disease clinically similar to types II and III, caused by defects in protein chaperoning[14] | KDELR2 | 619131 | autosomal recessive |
Sillence types[]
Sillence's four types have both a clinical and a genetic meaning; the descriptions below are clinical, and can be applied to a number of genetic types of OI. When used to refer to a genetic as well as a clinical type, it indicates that the clinical symptoms are indeed caused by mutations in the COL1A1 or COL1A2 genes which are inherited in an autosomal dominant fashion.[13]
Type I[]
Collagen is of normal quality but is produced in insufficient quantities.[8]: 1516 Bones fracture more easily than in the general public, but not as easily as more severe types of OI; there might be scoliosis, albeit mild compared to OI types III and IV, with a lower Cobb angle; the joints may be loose; blue sclerae may be apparent; hearing loss is likely to occur;[43]: Table 1 and, finally, there might be a slight decrease in height. Because cases exist missing one or more of these symptoms, OI type I in some cases goes undetected into adulthood.[8]: 1513–1514
Some further split type I into types I–A and I–B, defined as being distinguished by the absence (I–A) or presence (I–B) of dentinogenesis imperfecta (opalescent teeth).[43][44]: 217 People with type I generally have a normal lifespan.[45]
Type II[]
Collagen is fatally defective at its C-terminus.[8]: 1512 Most cases result in death shortly after birth, or within the first year of life, due to respiratory failure. Another common cause of death is intracranial bleeds from skull fractures present at, or sustained during or shortly after, birth.[8]: 1511 In many cases, the newborn already has multiple broken bones at the time of birth. Type II infants also exhibit severe respiratory problems, and have severely deformed bones. Sixty percent of infants die less than 24 hours after being born, and survival after the first year is extremely unlikely and normally requires mechanical ventilation.[46] In the rare cases of infants who survive their first year of life, severe developmental and motor delays are seen; neither of two infants studied in 2019, both aged around two years, had achieved head control, and both required a ventilator to breathe.[47]
Type II is also known as the "lethal perinatal" form of OI,[48] and is not compatible with survival into adulthood.[46] Due to similarly severely deformed bones, sometimes infants with severe type III are wrongly initially classified as type II; once long term survival is shown, they are considered as having type III instead.[8]: 1511
Type III[]
Collagen quantity is sufficient, but is not of a high enough quality.[8]: 1512 Clinical differentiation between types III and IV is not always simple, and is further confounded by the fact that an untreated adult with type IV may have worse symptoms than a treated adult with type III;[8]: 1511 [49] features only found in type III are its progressively deforming nature[8]: 1511–1512 and the presence of a face with a "triangular" appearance.[50] Another differentiating factor between type III and IV is blue sclerae; in type III, infants commonly have blue sclerae that gradually turn white with age, but blue sclerae are not seen in type IV.[1]: 294–296
OI type III causes osteopenic bones that fracture very easily, sometimes even in utero, often leading to hundreds of fractures during a lifetime;[51] early scoliosis that progresses until puberty; dwarfism (a final adult height frequently less than 4 feet or 120 centimetres); loose joints; and possible respiratory problems due to low rib cage volume causing low lung volumes.[8]: 1512
Due to the severity of the issues with the bones, neurological and seizure disorders are more likely to develop in type III.[8]: 1512 Basilar invagination, which puts pressure on the brainstem, may cause or contribute to early death; surgical treatment of it is more complex in OI cases.[8]: 1512 [11]: 106–107
Type IV[]
Collagen quantity is sufficient, but specifically α2 collagen is not of a high enough quality.[8]: 1512 Type IV is for cases of variable severity, which do not fit into either types III or I;[43] one of the required characteristics for type IV is having normal sclerae.[1]: 294–296 [33]: 114
In type IV, bone deformity is mild to severe, bones fracture easily (especially before puberty), dwarfism is common, vertebral collapse and scoliosis are evident, and hearing loss is possible,[6] although uncommon.[43][38] Type IV OI is mostly defined in contrast to type III and type I, being the clinical classification for patients somewhere in the middle ground between the two.[8]: 1511 As such, type IV OI is often termed "variable" OI,[33]: 111 with the severity of even those in the same family (so, with the same genetic mutation) differing.[43]
Prepubertal bone fracture rates are another way of clinically assessing type IV OI—those with it tend to have fracture rates of ≈1 per year, compared to ≈3 per year for severe OI (type III).[43]
As in type I, some further split type IV into types IV–A and IV–B, defined again by the absence (IV–A) or presence (IV–B) of dentinogenesis imperfecta.[43][44]: 217
Genetically defined types (types V–XXI)[]
As of 2020, fifteen types of OI are defined genetically:[13]
- Type V – Having the same clinical features as type IV, it can be clinically distinguished by observing a "mesh-like" appearance to a bone biopsy under a microscope. Type V can be further distinguished from other types of OI by the "V triad": an opaque band (visible on X-ray) adjacent to the growth plates; hypertrophic calluses (abnormally large masses of bony repair tissue) which form at fracture sites during the healing process; and calcification of the interosseous membrane of the forearm,[52] which may make it difficult to turn the wrist.[1]: 429
Other features of this condition may include pulled elbow, and, as in other types of OI, long bone bowing and hearing loss.[53]
Cases of this type are caused by mutations in the IFITM5 gene on chromosome 11p15.5.[53][41]
- Type VI – With the same clinical features as type III, it is distinguished by bones which have an appearance similar to that seen in osteomalacia.[1]: 168 Type VI is caused by a loss-of-function mutation in the SERPINF1 gene on chromosome 17p13.3.[1]: 170
- Type VII – OI caused by a mutation in the gene CRTAP on chromosome 3p22.3; clinically similar to OI types II and III, depending on affected individual. Type VII was the first recessive OI type confirmed, initially found among First Nations people in Quebec.[54][55]
- Type VIII – OI caused by a mutation in the gene LEPRE1 on chromosome 1p34.2; clinically similar to OI types II and III, depending on affected individual.[56]
- Type IX – OI caused by homozygous or compound heterozygous mutation in the PPIB gene on chromosome 15q22.31.[57]
- Type X – OI caused by homozygous mutation in the SERPINH1 gene on chromosome 11q13.[58]
- Type XI – OI caused by mutations in FKBP10 on chromosome 17q21. The mutations cause a decrease in secretion of trimeric procollagen molecules. Other mutations in this gene can cause autosomal recessive Bruck syndrome, which is similar to OI.[59]
- Type XII – OI caused by a frameshift mutation in SP7 on chromosome 12q13.13. This mutation causes bone deformities, fractures, and delayed tooth eruption.[60]
- Type XIII – OI caused by a mutation in the bone morphogenetic protein 1 (BMP1) gene on chromosome 8p21.3.[61] This mutation causes recurrent fractures, high bone mass, and hypermobile joints.[62]
- Type XIV – OI caused by mutations in the TMEM38B gene on chromosome 9q31.2. This mutation causes recurrent fractures and osteopenia, although the disease trajectory is highly variable.[63]
- Type XV – OI caused by homozygous or compound heterozygous mutations in the WNT1 gene on chromosome 12q13.12. It is autosomal recessive.[62]
- Type XVI – OI caused by mutations in the CREB3L1 gene on chromosome 11p11.2. The homozygous mutation causes prenatal onset of recurrent fractures of the ribs and long bones, demineralization, decreased ossification of the skull, and blue sclerae; it is clinically type II or type III.[64] Family members who are heterozygous for OI XVI may have recurrent fractures, osteopenia and blue sclerae.[64][65]
- Type XVII – OI caused by homozygous mutation in the SPARC gene on chromosome 5q33, causing a defect in the protein osteonectin, which leads to severe disease characterized by generalized platyspondyly, dependence on a wheelchair, and recurrent fractures.[66]
- Type XVIII – OI caused by homozygous mutation in the FAM46A gene on chromosome 6q14.1. Characterized by congenital bowing of the long bones, Wormian bones, blue sclerae, vertebral collapse, and multiple fractures in the first years of life.[67]
- Type XIX – OI caused by hemizygous mutation in the MBTPS2 gene on chromosome Xp22.12. Thus far, OI type XIX is the only known type of OI with an X-linked recessive pattern of inheritance, making it the only type that is more common in males than females. OI type XIX disrupts regulated intramembrane proteolysis, which is critical for healthy bone formation.[68]
- Type XX – OI caused by homozygous mutation in the MESD gene on chromosome 15q25.1. Initial studies of type XX indicate that it may cause global developmental delay, a first among OI types. OI type XX disrupts the Wnt signaling pathway, which is thought to have a role in bone development.[42]
- Type XXI – OI caused by homozygous mutation in the KDELR2 gene on chromosome 7p22.1. Causes disease clinically similar to types II and III, thought to be related to inability of chaperone protein HP47 to unbind from collagen type I, as to do so it needs to bind to the missing ER lumen protein retaining receptor 2 protein encoded by KDELR2.[14]
Given the rapid rate of type discovery, it is extremely likely that there are other genes associated with OI that have yet to be reported.
Genetics[]

Osteogenesis imperfecta is a group of genetic disorders, all of which cause bone fragility. OI has high genetic heterogeneity, that is, many different genetic mutations lead to the same or similar sets of observable symptoms (phenotypes).[69]
The main causes for developing the disorder are a result of mutations in the COL1A1 and/or COL1A2 genes which are jointly responsible for the production of collagen type I.[70] Approximately 90% of people with OI are heterozygous for mutations in either the COL1A1 or COL1A2 genes.[71] There are several biological factors that are results of the dominant form of OI. These factors include: intracellular stress; abnormal tissue mineralization; abnormal cell to cell interactions; abnormal cell-matrix interactions; a compromised cell matrix structure; and, abnormal interaction between non-collagenous proteins and collagen.[72]
Previous research lead to the belief that OI was an autosomal dominant disorder with few other variations in genomes.[73] However, with the lowering of the cost of DNA sequencing in the wake of 2003's Human Genome Project,[74] autosomal recessive forms of the disorder have been identified.[75] Recessive forms of OI relate heavily to defects in the collagen chaperones responsible for production of procollagen and the assembly of the related proteins.[76] Examples of collagen chaperones that are defective in patients with recessive forms of OI include chaperone HSP47 (Cole-Carpenter syndrome) and FKBP65.[35] Mutations in these chaperones result in an improper folding pattern in the collagen 1 proteins which causes the recessive form of the disorder.[35] There are three significant types of OI that are a result of mutations in the collagen prolyl 3-hydroxylation complex (components CRTAP, P3H1, and CyPB).[35] These components are responsible for the modification of collagen α1(l)Pro986.[35] Mutations in other genes such as SP7, SERPINF1, TMEM38B and BMP1 can also lead to irregularly formed proteins and enzymes that result in other recessive types of osteogenesis imperfecta.[35]
Defects in the proteins pigment epithelium-derived factor (PEDF) and bone-restricted interferon-induced transmembrane protein (BRIL) are the causes of type V and VI osteogenesis imperfecta.[77] Defects in these proteins lead to defective bone mineralization which causes the characteristic brittle bones of osteogenesis imperfecta.[77] A single point mutation in the 5′ untranslated region (5′ UTR) of the IFITM5 gene, which encodes BRIL, is linked directly to OI type V.[53][78]
In the rare case of type XIX, first discovered in 2016, OI is inherited as an X-linked genetic disorder, with its detrimental effects resulting ultimately from a mutation in the gene MBTPS2.[68] Genetic research is ongoing, and it is uncertain when all the genetic causes of OI will be identified, as the number of genes that need to be tested to rule out the disorder continue to increase.
In a study of 37 families, a 1.3% chance was found that OI recurs in multiple siblings born to two unaffected parents—this is a much higher rate than would be expected if all such recurrences were de novo.[79] The cause is genetic mosaicism; that is, some of, or most of, the germ cells of one parent have a dominant form of OI, but not enough of their somatic cells do to cause symptoms or obvious disability in the parent—the parent's different cells have two (or more) sets of slightly different DNA.[79][8]: 1513 It has been clinically observed that ≈5–10% of cases of OI types II and III are attributable to genetic mosaicism.[1]: 532
Pathophysiology[]
People with OI are born with defective connective tissue, or without the ability to make it, usually because of a deficiency of type I collagen.[80] This deficiency arises from an amino acid substitution of glycine to a bulkier amino acid, such as adenine, in the collagen protein's triple helix structure. The larger amino acid side-chains lead to steric effects that creates a bulge in the collagen complex, which in turn influences both the molecular nanomechanics and the interaction between molecules, which are both compromised.[81] As a result, the body may respond by hydrolyzing (destroying) the improper collagen structure. If the body does not hydrolize the improper collagen, the relationship between the collagen fibrils and hydroxyapatite crystals to form bone is altered, causing brittleness.[82] Bone fractures occur because the stress state within collagen fibrils is altered at the locations of mutations, where locally larger shear forces lead to rapid failure of fibrils even at moderate loads, because the homogeneous stress state normally found in healthy collagen fibrils is lost.[81] OI is therefore a multi-scale phenomenon, where defects at the smallest levels of tissues (genetic, nano, micro) domino to affect the macro level of tissues.
Diagnosis[]
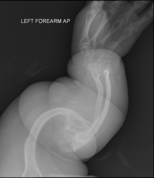
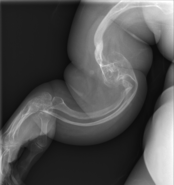
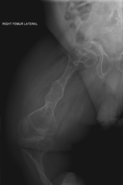
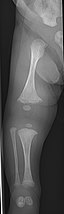
Diagnosis is typically based on medical imaging, including plain X-rays, and symptoms. Signs on medical imaging include abnormalities in all extremeties and the spine.[83]
An OI diagnosis can be confirmed through DNA or collagen testing, but in many cases, the occurrence of bone fractures with little trauma and the presence of other clinical features such as blue sclerae are sufficient for a diagnosis. A skin biopsy can be performed to determine the structure and quantity of type I collagen. DNA testing can confirm the diagnosis, however, it cannot exclude it because not all mutations causing OI are yet known and/or tested for. OI type II is often diagnosed by ultrasound during pregnancy, where already multiple fractures and other characteristic features may be visible. Relative to control, OI cortical bone shows increased porosity, canal diameter, and connectivity in micro-computed tomography.[84] Severe types of OI can usually be detected before birth by using an in vitro genetic testing technique.[85]
Genetic testing[]
In order to determine whether osteogenesis imperfecta is present, genetic sequencing of the most common problematic genes, COL1A1, COL1A2, and IFITM5, may be done;[86] if no mutation is found yet OI is still suspected, the other 10+ genes known to cause OI may be tested.[13] Duplication and deletion testing is also suggested to parents who suspect their child has OI.[86] The presence of frameshift mutations caused by duplications and deletions is generally the cause of increased severity within the disease.[86]
Differential diagnosis[]
An important differential diagnosis of OI is child abuse, as both may present with multiple fractures in various stages of healing.[8]: 1514 Differentiating them can be difficult, especially when no other characteristic features of OI are present.
Other differential diagnoses include rickets and osteomalacia, both caused by malnutrition, as well as rare skeletal syndromes as Bruck syndrome, hypophosphatasia, geroderma osteodysplasticum, and Ehlers–Danlos syndrome.[8]: 1513 [1]: 253–256 Various forms of osteoporosis, such as iatrogenic osteoporosis, idiopathic juvenile osteoporosis, disuse osteoporosis and exercise-related osteoporosis should also be considered as explanations when OI is suspected.[1]: 255–256
Treatment[]
There is no cure for osteogenesis imperfecta.[6] Maintaining a healthy lifestyle by exercising and avoiding smoking can help prevent fractures. Treatment may include care of broken bones, pain medication, physical therapy, mobility aids such as braces or wheelchairs, and surgery.
Acute bone fracture care[]
Bone fractures are treated in individuals with osteogenesis imperfecta in much the same way as they are treated in the general population—OI bone heals at the same rate as non-OI bone.[1]: 431 A greater emphasis is placed on using lightweight materials to immobilize the fracture, as in moderate or severe types of OI, using heavy casts, such as hip spica casts, can cause fractures at the bones at the boundaries of the cast, as well as generalized osteopenia.[1]: 431 The lightweight cast or splint is then replaced with a removable orthosis after a few weeks and once evidence of union is seen on X-ray.[1]: 431 In order to prevent a nonunion or malunion, all fractures should be immobilized, even if the fracture seems trivial (microfracture),[1]: 439 as people with OI are at greater risk of nonunion.[1]: 438
Bone infections secondary to fractures are treated as and when they occur with the appropriate antibiotics and antiseptics, as in the general population.[1]: 424
Bisphosphonates[]
In 1998, an initial observational trial demonstrated the effectiveness of intravenous pamidronate, a bisphosphonate which had previously been used in adults to treat osteoporosis. In severe OI, this trial showed that pamidronate reduced bone pain, prevented new vertebral fractures, reshaped previously fractured vertebral bodies, and reduced the number of long-bone fractures.[87]
Although oral bisphosphonates are more convenient and cheaper, they are not absorbed as well, and intravenous bisphosphonates are generally more effective, although this is under study. Some studies have found oral and intravenous bisphosphonates, such as oral alendronate and intravenous pamidronate, equivalent.[88] In a 2013 double-blind trial of children with mild OI, oral risedronate increased bone mineral densities, and reduced nonvertebral fractures. However, it did not decrease new vertebral fractures.[89][90] A Cochrane review in 2016 concluded that though bisphosphonates seem to improve bone mineral density, it is uncertain whether this leads either to a reduction in bone fractures or improvement in the quality of life of individuals with osteogenesis imperfecta.[7] Even in trials with as many as 125 children, no causal link has been found between bisphosphonates and decreased fracture rates; placebo controlled trials were also unable to prove that they brought about increased strength, motor control or lower pain levels.[72]
Bisphosphonates are not as effective at increasing the bone mineral density of adults.[17]
Surgery[]
A surgery of any type inherently carries more risks when done on a patient who has (especially moderate to severe) OI. Skeletal deformities and dentinogenesis imperfecta may hinder access to the airway.[1]: 333 Use of, and weaning off of, mechanical ventilation is also more challenging to carry out on patients with OI.[1]: 333 During the procedure itself or the healing process, defective OI collagen may lead to bleeding diatheses.[1]: 333
The safety of anesthesia is also of more concern among patients with OI,[1]: 333 with anesthetic complications 5.6x more likely to occur when the patient has OI type III.[91] A unique concern of anesthesia in OI is perioperative fracture—fractures sustained due to patient transfer and airway access techniques that, while routine when a patient's bones are strong, may cause injury with brittle OI bones.[92] As an example, due to a 1972 report of a humerus fracture from a sphygmomanometer cuff sustained in an OI patient during surgery, blood pressure monitoring protocols are often modified for patients with OI, with neonatal size cuffs and machine settings being used even in adults;[93]: ¶11.72 further, the least deformed of the patient's limbs is preferred to receive the cuff.[94]: ¶14.23
Rodding[]
Metal rods can be surgically inserted in the long bones to improve strength, a procedure developed by Harold A. Sofield when he was Chief of Staff at Chicago's Shriners Hospitals for Children, a hospital that offers orthopedic care and surgery to children regardless of their family's ability to pay.[95] Large numbers of children with OI came to Shriners, and Sofield experimented with various methods to strengthen their bones.[96] In 1959, with Edward A. Millar [sic], Sofield wrote a seminal article describing a three-part surgery that seemed radical at the time: precisely breaking the bones ("fragmentation"), putting the resulting bone fragments in a straight line ("realignment"), then placing metal rods into the intramedullary canals of the long bones to stabilize and strengthen them ("rod fixation").[97] His treatment proved useful for increasing the mobility of people with OI, and it has been adopted throughout the world—it became standard surgical treatment for severe OI by 1979, in which year David Sillence found that ≈2⁄3 of the patients he surveyed with OI type III had undergone at least one rodding surgery.[33]: 108
Rodding surgery is often done with the hope that it will offer a path to ambulation, walking, to patients with moderate or severe OI. A 2020 review in The Journal of Bone and Joint Surgery (JB&JS) found it remains broadly popular: ≈2⁄3 of people with OI types III and IV (severe OI) have undergone some form of rodding surgery in their lives, at a mean age of 41⁄10 and 71⁄2 years respectively;[18]: Table I one possible explanation for a tendency towards earlier intervention in type III is that one half of affected children could not walk at all without the surgery, as their limbs were more bowed, so surgery was sought sooner.[18]
In those with type III OI who had undergone rodding surgery, 79.5% had the femurs and tibias of both legs rodded.[18]: Table I The most common form of rods used are intramedullary (IM) rods, some of which, such as the Fassier–Duval IM rod, are telescoping, meaning that they are designed to grow as the child grows, in an attempt to avoid the necessity of revision surgeries.[98] Telescoping IM rods are widely used,[99] and the common Fassier–Duval IM rod is designed to be used to rod the femur, tibia, and humerus.[100]: 1 The surgery involves breaking the long bones in between one and three (or more)[99]: Figure 4 places, then fixing the rod alongside the bone to keep it straight.[100]: 11
While telescoping IM rods are intended to grow along with both the femur and tibia in developing children; surgeons have a preference to use non-telescoping IM rods, such as Rush rods, in the tibia, which grows less comparatively—the JB&JS review found that while 69.7% of femurs were treated with telescoping IM rods, only 36.9% of tibiae were.[18]: Table IV
While the review in the JB&JS was able to correlate receiving rodding surgery with greater mobility across all types of OI, in patients with type IV, the surgery did not decrease the incidence of broken bones as compared to non-rodded patients—while type IV patients with rodded tibiae experienced 0.93 tibia fractures per year, patients with natural tibiae experienced only 0.81. However, in patients with type III, rodding surgery decreased the average number of tibia fractures per year from 0.84 to 0.57.[18]: Table V
Spinal[]
Spinal fusion can be performed either as a preventative measure or to correct existing scoliosis, although the inherent fragility of OI bone makes this operation more complex in OI patients than it does with patients who have adolescent idiopathic scoliosis, but normal bone density.[101] Despite the risks, however, three Nemours–duPont orthopedic surgeons who specialize in surgical intervention for osteogenesis imperfecta recommend operating if the curve is greater than 50° after a child is past peak height velocity, as the spine's curve can continue to worsen even into adulthood.[11]: 104
Due to the risk involved, the same surgeons recommend that surgery for basilar impressions and basilar invaginations should only be carried out if the pressure being exerted on the spinal cord and brain stem is causing actual neurological symptoms.[11]: 106–107 Once basilar invagination has become symptomatic, only surgery can halt or reverse the progression of neurological deficits.[1]: 345
Physical therapy[]
Physical therapy is generally recommended, however individualized protocols are required due to the variability of OI.[1]: 378 Physical therapy is used to strengthen muscles, improve motility, improve flexibility, and help with weight maintenance, although it must be done in a gentle manner to minimize the risk of bone fracture.[1]: 378 In people with OI, exercise often involves water aerobics, light resistance exercises, and walking, if the patient is able.[1]: 378 However, even in patients with mild OI, contact sports, as well as activities likely to put unnecessary stress on the joints, such as jumping, are contraindicated due to the risks they pose.[1]: 378
Individuals with more limited mobility are encouraged to change positions regularly throughout the day; people who sit in a wheelchair most or all of the day are recommended to get out of it every two hours, as a form of exercise, to decrease stiffness, and to prevent pressure ulcers.[1]: 378
Individuals with moderate to severe OI, who require assistive mobility devices and adapted vehicles, face significant barriers to access wheelchair-accessible pools or gyms—they either may not have any in their area, nor the means to get there.[1]: 378 Obesity may be more likely to present among those with severe OI, (especially after the age of 20,) and can, in some, cause further declines in mobility.[102][1]: 371, 373
Tilt table whole body vibration may also be done to increase the mobility of long-term immobilized (bedridden) patients with OI; in at least two cases it helped bedridden children to be able to sit upright.[103][72]
Teeth[]
More than 1 in 2 people with OI also have dentinogenesis imperfecta (DI)—a congenital abnormality in the formation of dentin, one of the four major components of the human tooth.[104] Dental treatment may pose as a challenge as a result of the various deformities, skeletal and dental, due to OI. Children with OI should go for a dental check-up as soon as their teeth erupt; this may minimize tooth structure loss as a result of abnormal dentine, and they should be monitored regularly to preserve their teeth and oral health.[104]
Many people with OI are treated with bisphosphonates, and there are several possible related complications with dental procedures, for example, medication-related osteonecrosis of the jaw (MRONJ). However, no report of bisphosphonate-related MRONJ in either a child or adult with OI was found in a 2016 Cochrane review of the safety and efficacy of bisphosphonates for OI.[7]
Prevention[]
As a genetic disorder, the mainstay of twenty-first century prevention of osteogenesis imperfecta is based on preventing affected individuals from being born in the first place. Genetic counseling can help patients and their families determine what types of screening, if any, are right for their situation. Patients can consider preimplantation genetic diagnosis after in vitro fertilization to select fertilized embryos which are not affected.[8]: 247–248 Common mutations which cause OI may be caught by exome sequencing and whole genome sequencing. If a pregnancy is already in progress, the procedure of amniocentesis may be undergone to see if the fetus is affected.[8]: 247 If affected, it is up to the family to consider whether or not they want to terminate the pregnancy and try again—raising questions of medical ethics and a woman's right to choose.[105][106]
Without intervention, patients with the most common mutations causing osteogenesis imperfecta have a 50% chance per gestation of passing on the disorder, as these mutations are inherited in an autosomal dominant pattern of Mendelian inheritance.[8]: 247 Those with the rare autosomal recessive forms of OI have a 25% chance of passing on the disorder. Genetic testing of the affected members of the family can be used to determine which inheritance pattern applies.[8]: 101
As OI type I may be difficult to detect in a newborn child, the cord blood of the child can be tested to determine if it has been passed on, if the family has already rejected the more invasive genetic screening methods.[8]: 247 In more severe cases, the diagnosis may be able to be done via ultrasound, especially if OI is already a possibility.[8]: 248 An ethical concern with prenatal screening for OI often arises when parents inquire as to how severely affected their child will be—such questions are as yet difficult to answer conclusively.[8]: 382
If a non-affected person has already had a child with OI, there is a greater likelihood, (although still quite remote,) that their future children will have OI due to genetic mosaicism.[79][8]: 100, 1513
The disability rights critique of prenatal screening for OI, held by some bioethicists and some affected individuals, negatively compares it to eugenics, with even those not opposed to abortion opposing selective abortions on the ethical ground that their existence betrays the belief that the lives of those with OI are "less worth living [and] less valuable".[8]: 388
Prognosis[]
The prognosis of osteogenesis imperfecta depends entirely on its type (see § Classification).
Life expectancy[]
In the mild form of the disorder, type I, the life expectancy of patients is near that of the general population.[1]: 461 In type II, however, patients only very rarely live past the age of two, and typically die in their first weeks of life.[8]: 1511 Assessment of the life expectancy of patients with types III and IV is more complicated, as lifestyle choices can cause fatally traumatic injuries that would not have otherwise occurred, or not been fatal in the general population. Life expectancy in type IV OI is thought to be close to normal, but in type III it is lower than in the general population.[40]
A 2016 study of data in Denmark's found that across all types of OI, all-cause mortality was three times higher, leading to a loss of around seven years in females and nine years in males.[12] A 1996 study published in the British Medical Journal found that mortality in type III OI is significantly higher, with many patients dying in their 20's, 30's, and 40's; patients who survive to the age of 10 were further found to have longer life expectancy than newborns.[107]
Mobility[]
People with mild (type I) OI as adults need few pieces of adaptive equipment, although in infancy they reach motor milestones at a significant delay compared to the general population.[1]: 477
With adaptive equipment such as crutches, motorized wheelchairs, splints, reach extenders, and/or modifications to the home, many individuals with moderate to severe OI can achieve or maintain a significant degree of independence.[15][1]: 488 With treatment and physical therapy, the maximum levels of mobility are expected to be unassisted community walking for type I, household or exercise walking for type III, and household or community walking for type IV; due to the variability of OI between individuals, mobility achieved varies and may be below this expected maximum.[1]: 476
Epidemiology[]
In the United States, the incidence of osteogenesis imperfecta is estimated to be one per 20,000 live births.[108] An estimated 20,000 to 50,000 people are affected by OI in the United States. The most common types are I, II, III and IV, while the rest are very rare.[109] Most people with OI receive it from a parent, but in many cases it is a brand new (de novo or "sporadic") mutation in a family. Among a study of patients with survivable types of OI, OI type III is most often de novo (85%), followed by type IV (50%) and type I (34%).[37]: Table 1
Some populations can have a higher incidence of OI than would be otherwise expected if they have a larger than average number of carriers of the recessive forms of the disease.[1]: 20–21 [110]
History[]
The condition, or types of it, has had various other names over the years and in different nations; "osteogenesis imperfecta" has, however, been the most widely accepted name for the condition since the late 20th century. Among some of the most common alternatives are "fragilitas ossium";[111] "Ekman–Lobstein syndrome", and "Vrolik syndrome", both eponyms; and, the colloquialism, "brittle bone disease".[1]
Earliest recorded cases[]
OI has been identified in an ancient Egyptian infant mummified in around 1000 BC, originally dismissed by archaeologists as containing the remains of a monkey.[112][113]: 161 The Norse king Ivar the Boneless, who lived c. 800 CE,[114] is speculated to have had OI, as well.[115]
Nicolas de Malebranche is oft credited as being the first person to describe the physical characteristics of OI in his 1688 book , in which he describes a man who has had his "bones broken in the places a murderer's would be" all his life. His confident description of the pathology of the disorder, however, which creates what he termed «enfants monstrueux» ("monstrous children"), is scientifically void—he wrote that it was due to the mother's antepartum viewership of a public execution by breaking wheel.[20]: 683 [113]: 165–168 [116]
The earliest modern scientific studies of OI began in 1788 by Olof Jakob Ekman, who described the condition, which he termed "osteomalacia congenital", in his doctoral thesis and mentioned cases of it going back to 1678, all in the same family, through three generations.[117] Ekman's description of the condition mentioned dwarfism, bone fragility, and bowing of the long bones.[118]: 763 In 1831, Edmund Axmann gave a detailed description of it in himself and his two brothers, being the first to mention blue sclerae as a characteristic sign of OI.[20]: 683 Jean Lobstein first described the mild form of the condition, today known as type I, in 1833, calling it "osteopsathyrosis idiopathica".[3]: 347
It was not until 1912 that hearing loss was positively recognized as a symptom of OI, first mentioned in a brief paper by the English physician Charles Allen Adair-Dighton.[119][113]: 168–169
Of the term[]
Willem Vrolik, a Dutch anatomist who was also curator of the "Museum Vrolikianum", which made him privy to many specimens of bodies having birth defects, coined the term "osteogenesis imperfecta"[20]: 683 in his bilingual Latin and Dutch language book on teratology, Tabulae ad illustrandam embryogenesin hominis, tam naturalem quam abnormem, first published in 1849.[120]
Included is a description of the remains of an infant who had what is now known as perinatally fatal OI type II[3]: 347 [1]: 5 (as verified in a 1998 re-examination of the remains by Baljet et al.).[121] The remains were first given to Vrolik's father, who could not make sense of them. Vrolik described poorly mineralized bones, bowed long bones, and fractures in various states of healing.[122] Vrolik correctly determined that what he termed OI in the infant was not caused by secondary rickets, but a congenital abnomality causing primary osteopenia; he theorized this was due to a lack of "intrinsic generative energy".[121]
Of its classification[]
Classification of OI has also evolved as scientific understanding of it has improved. Before the advent of modern genetic testing, OI was classified in two broad groups: osteogenesis imperfecta congenita, and osteogenesis imperfecta tarda, a division first proposed by the German physician E. Looser in 1906.[111][123] Congenita was used to describe the modern clinical types II, III, and some cases of IV, where upon birth the condition was obvious, either due to bowing of the limbs or due to fractures sustained in utero.[33] Tarda was used to classify the modern OI type I and some cases of type IV, where the inherent fragility of the bones did not become clear until long after birth.[111] The idea that these "late" and "prenatal" forms were manifestations of the same disorder was first proposed in 1897 by Martin Benno Schmidt;[124] by the 1950's this fact was well accepted.[3]: 346
The modern system of four types (I, II, III, IV), meanwhile, were introduced in a paper by David Sillence, Alison Senn, and David Danks in the Journal of Medical Genetics in 1979,[33][125] and have since become standard terms among doctors, patients, and researchers.[34][43][126] The modern genetic types, (those with numbers greater than IV,) have come into use as more and more recessively inherited forms of OI have been discovered since the discovery of the first one by Roy Morello et al. in 2006.[38][73][54] In 2010, the International Nomenclature Group for Constitutional Disorders of the Skeleton (INCDS) "freed" the Sillence types from molecular reference, acquiescing to their new clinical-first role in the wake of what was to them a "surprising" increase in the number of genetic causes of OI.[126][43]
Society and culture[]
Other animals[]
In dogs, OI is an autosomal recessive condition, meaning that dogs with two copies of the allele will be affected.[127] Many breed organizations and veterinarians offer OI tests to tell if a dog is a carrier of OI.[127][128] Dogs who are heterozygous for OI should only be bred to non-carriers. Homozygous carriers should never be bred, unless it is to a non-carrier.[128]
Naturally occurring mutations causing OI have been found in Golden Retrievers, Dachshunds, and Beagles. OI has also been identified in zebrafish and mice.[129]
Although dogs, mice, fish, and humans are not genetically identical, some of these animal models have been officially recognized to represent the varying types of OI in humans. For example, homozygous oim/oim mice experience spontaneous bone fractures, small body size, and kyphosis, making them a model of OI type III. Meanwhile, heterozygous oim/+ mice appear normal but have bones which are quite a bit weaker than wild mice, making them a model for OI type I.[129][130] As in human OI, the location on the gene which is mutated affects the severity of resulting disease—the G859C Col1a1 mouse is a model for OI type II as affected mice all die in the perinatal period.[129]
Animal testing on identified animal models may lead to human therapies for OI.[129]
See also[]
References[]
- ^ Jump up to: a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am an ao ap aq ar as at au av aw ax ay az Shapiro JR, Byers PH, Glorieux FH, Sponseller PD (2014). Osteogenesis imperfecta: a translational approach to brittle bone disease (2nd ed.). London: Elsevier Inc. ISBN 978-0-12-397165-4. OCLC 876364090.
- ^ Jump up to: a b c d e f g h i j k "Osteogenesis imperfecta". Genetics Home Reference. U.S. National Library of Medicine, National Institutes of Health. 18 August 2020. Archived from the original on 15 September 2021. Retrieved 15 August 2021.
- ^ Jump up to: a b c d e Davel JG, Fichardt T, Van Der Spuy D (October 1956). "Osteogenesis imperfecta". Archives of Disease in Childhood. 31 (159): 346–353. doi:10.1136/adc.31.159.346. PMC 2011939. PMID 13363481.
- ^ Jump up to: a b "osteogenesis". Lexico UK Dictionary. Oxford University Press. Retrieved 23 August 2021. "osteogenesis imperfecta". Lexico UK Dictionary. Oxford University Press. Retrieved 23 August 2021.
- ^ Jump up to: a b "osteogenesis". Merriam-Webster Dictionary. Retrieved 23 August 2021. "osteogenesis imperfecta". Merriam-Webster Dictionary. Retrieved 23 August 2021.
- ^ Jump up to: a b c d e f g h i j k l m n o Lee B, Krakow D (July 2019). "Osteogenesis Imperfecta Overview". National Institute of Arthritis and Musculoskeletal and Skin Diseases. National Institutes of Health. Archived from the original on 9 May 2021. Retrieved 21 August 2021.CS1 maint: uses authors parameter (link)
- ^ Jump up to: a b c d e f g h i j k Dwan K, Phillipi CA, Steiner RD, Basel D (October 2016). "Bisphosphonate therapy for osteogenesis imperfecta". The Cochrane Database of Systematic Reviews. 10: CD005088. doi:10.1002/14651858.CD005088.pub4. PMC 6611487. PMID 27760454 – via National Center for Biotechnology Information.
- ^ Jump up to: a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae Rowe DW (2008). "Osteogenesis imperfecta". Principles of bone biology (3rd ed.). Amsterdam: Elsevier. pp. 1511–1531. ISBN 978-0-12-373884-4. OCLC 267135745.
Dentinogenesis imperfecta (DI) is most frequent in OI types III and IV, and overall, affects about 15% of OI patients among the different phenotypes.
- ^ McNeeley MF, Dontchos BN, Laflamme MA, Hubka M, Sadro CT (December 2012). "Aortic dissection in osteogenesis imperfecta: case report and review of the literature". Emergency Radiology. 19 (6): 553–536. doi:10.1007/s10140-012-1044-1. PMID 22527359. S2CID 11109481.
- ^ Jump up to: a b c d LoMauro A, Pochintesta S, Romei M, D'Angelo MG, Pedotti A, Turconi AC, et al. (27 April 2012). "Rib Cage Deformities Alter Respiratory Muscle Action and Chest Wall Function in Patients with Severe Osteogenesis Imperfecta". PLOS ONE. 7 (4): e35965. doi:10.1371/journal.pone.0035965. ISSN 1932-6203. PMC 3338769. PMID 22558284.
- ^ Jump up to: a b c d e f Wallace MJ, Kruse RW, Shah SA (February 2017). "The Spine in Patients With Osteogenesis Imperfecta". The Journal of the American Academy of Orthopaedic Surgeons. 25 (2): 100–109. doi:10.5435/JAAOS-D-15-00169. PMID 28009707.
- ^ Jump up to: a b c Folkestad L, Hald JD, Canudas-Romo V, Gram J, Hermann AP, Langdahl B, et al. (2016). "Mortality and Causes of Death in Patients With Osteogenesis Imperfecta: A Register-Based Nationwide Cohort Study". Journal of Bone and Mineral Research. 31 (12): 2159–2166. doi:10.1002/jbmr.2895. ISSN 1523-4681.
- ^ Jump up to: a b c d e Marom R, Rabenhorst BM, Morello R (October 2020). "Osteogenesis imperfecta: an update on clinical features and therapies". European Journal of Endocrinology. 183 (4): R95–R106. doi:10.1530/EJE-20-0299. PMC 7694877. PMID 32621590.
- ^ Jump up to: a b c d Van Dijk FS, Semler O, Etich J, Köhler A, Jimenez-Estrada JA, Bravenboer N, et al. (5 November 2020). "Interaction between KDELR2 and HSP47 as a Key Determinant in Osteogenesis Imperfecta Caused by Bi-allelic Variants in KDELR2". The American Journal of Human Genetics. 107 (5): 989–999. doi:10.1016/j.ajhg.2020.09.009. ISSN 0002-9297. PMC 7675035. PMID 33053334.
- ^ Jump up to: a b Donohoe M (2020), Kruse RW (ed.), "Therapy, Orthotics and Assistive Devices for Osteogenesis Imperfecta", Osteogenesis Imperfecta: A Case-Based Guide to Surgical Decision-Making and Care, Springer International Publishing, pp. 21–37, doi:10.1007/978-3-030-42527-2_3, ISBN 978-3-030-42526-5
- ^ Harrington J, Sochett E, Howard A (December 2014). "Update on the evaluation and treatment of osteogenesis imperfecta". Pediatric Clinics of North America. 61 (6): 1243–1257. doi:10.1016/j.pcl.2014.08.010. PMID 25439022.
- ^ Jump up to: a b Chevrel G, Schott AM, Fontanges E, Charrin JE, Lina-Granade G, Duboeuf F, et al. (February 2006). "Effects of oral alendronate on BMD in adult patients with osteogenesis imperfecta: a 3-year randomized placebo-controlled trial". Journal of Bone and Mineral Research. 21 (2): 300–306. doi:10.1359/JBMR.051015. PMID 16418786. S2CID 34089615.
- ^ Jump up to: a b c d e f Rodriguez Celin M, Kruger KM, Caudill A, Nagamani SC, Harris GF, Smith PA (11 September 2020). "A Multicenter Study of Intramedullary Rodding in Osteogenesis Imperfecta". JB & JS Open Access. 5 (3): e20.00031. doi:10.2106/JBJS.OA.20.00031. PMC 7489747. PMID 32984750.
- ^ Jump up to: a b Kelly EB (2012). Encyclopedia of Human Genetics and Disease. ABC-CLIO. p. 613. ISBN 9780313387135. Archived from the original on 5 November 2017.
- ^ Jump up to: a b c d e f Dang Do AN, Marini JC (27 January 2021). "Osteogenesis imperfecta". In Carey JC, Cassidy SB, Battaglia A, Viskochil D (eds.). Cassidy and Allanson's Management of Genetic Syndromes. John Wiley & Sons. pp. 683–705. ISBN 978-1-119-43267-8.
- ^ Johnson MT, Morrison S, Heeger S, Mooney S, Byers PH, Robin NH (February 2002). "A variant of osteogenesis imperfecta type IV with resolving kyphomelia is caused by a novel COL1A2 mutation". Journal of Medical Genetics. 39 (2): 128–132. doi:10.1136/jmg.39.2.128. PMC 1735034. PMID 11836364.
- ^ Atta I, Iqbal F, Lone SW, et al. "Journal of the College of Physicians and Surgeons Pakistan" (PDF). Journal of the College of Physicians and Surgeons Pakistan. 24 (9): 653–657.
- ^ Cabral WA, Makareeva E, Colige A, Letocha AD, Ty JM, Yeowell HN, et al. (1 May 2005). "Mutations Near Amino End of α1(I) Collagen Cause Combined Osteogenesis Imperfecta/Ehlers-Danlos Syndrome by Interference with N-propeptide Processing". Journal of Biological Chemistry. 280 (19): 19259–19269. doi:10.1074/jbc.m414698200. ISSN 0021-9258.
- ^ Rasmussen SA (2 December 2020). "Combined osteogenesis imperfecta and Ehlers–Danlos syndrome 1; OIEDS1". Online Mendelian Inheritance in Man. Johns Hopkins University. 619115. Retrieved 17 August 2021.CS1 maint: uses authors parameter (link)
- ^ Jump up to: a b Peterson CR, Monk EA, McAllion SJ (1 April 2001). "How common is hearing impairment in osteogenesis imperfecta?". The Journal of Laryngology & Otology. 115 (4): 280–282. doi:10.1258/0022215011907442. ISSN 0022-2151. PMID 11276328. S2CID 12303464.
- ^ Vernick D (2 November 2005). "OI Issues: Hearing Loss". Archived from the original on 20 January 2012. Retrieved 4 November 2018.
- ^ Santos F, McCall AA, Chien W, Merchant S (December 2012). "Otopathology in Osteogenesis Imperfecta". Otology & Neurotology. 33 (9): 1562–1566. doi:10.1097/MAO.0b013e31826bf19b. PMC 3498599. PMID 22996160.
- ^ Kuurila K, Grénman R, Johansson R, Kaitila I (July 2000). "Hearing loss in children with osteogenesis imperfecta". European Journal of Pediatrics. 159 (7): 515–619. doi:10.1007/s004310051322. PMID 10923226. S2CID 8729406.
- ^ Jump up to: a b Khandanpour N, Connolly DJ, Raghavan A, Griffiths PD, Hoggard N (1 December 2012). "Craniospinal abnormalities and neurologic complications of osteogenesis imperfecta: imaging overview". Radiographics. 32 (7): 2101–2112. doi:10.1148/rg.327125716. PMID 23150860.
- ^ Violas P, Fassier F, Hamdy R, Duhaime M, Glorieux FH (September 2002). "Acetabular protrusion in osteogenesis imperfecta". Journal of Pediatric Orthopedics. 22 (5): 622–625. doi:10.1097/01241398-200209000-00010. PMID 12198464. S2CID 37895736.
- ^ Jump up to: a b Lee JH, Gamble JG, Moore RE, Rinsky LA (September 1995). "Gastrointestinal problems in patients who have type-III osteogenesis imperfecta". The Journal of Bone and Joint Surgery. American Volume. 77 (9): 1352–1356. doi:10.2106/00004623-199509000-00010. PMID 7673285.
- ^ Turkalj M, Miranović V, Lulić-Jurjević R, Gjergja Juraški R, Primorac D (25 September 2017). "Cardiorespiratory complications in patients with osteogenesis imperfecta". Paediatria Croatica. 61 (3): 106–111. doi:10.13112/PC.2017.15. ISSN 1330-1403.
- ^ Jump up to: a b c d e f Sillence DO, Senn A, Danks DM (April 1979). "Genetic heterogeneity in osteogenesis imperfecta". Journal of Medical Genetics. 16 (2): 101–116. doi:10.1136/jmg.16.2.101. PMC 1012733. PMID 458828.
- ^ Jump up to: a b c d Van Dijk FS, Pals G, Van Rijn RR, J Nikkels PG, Cobben JM (1 January 2010). "Classification of Osteogenesis Imperfecta revisited". European Journal of Medical Genetics. 53 (1): 1–5. doi:10.1016/j.ejmg.2009.10.007. ISSN 1769-7212.
In 1979 Sillence proposed a classification of Osteogenesis Imperfecta (OI) in OI types I, II, III and IV. In 2004 and 2007 this classification was expanded with OI types V–VIII because of distinct clinical features and/or different causative gene mutations.
- ^ Jump up to: a b c d e f Marini JC, Blissett AR (August 2013). "New genes in bone development: what's new in osteogenesis imperfecta". The Journal of Clinical Endocrinology and Metabolism. 98 (8): 3095–3103. doi:10.1210/jc.2013-1505. PMC 3733862. PMID 23771926.
- ^ McKusick VA, Hamosh A (23 October 2018). "Osteogenesis imperfecta, type I; OI1". Online Mendelian Inheritance in Man. Johns Hopkins University. 166200. Retrieved 17 August 2021.
- ^ Jump up to: a b c d e Zhytnik L, Maasalu K, Duy BH, Pashenko A, Khmyzov S, Reimann E, et al. (March 2019). "De novo and inherited pathogenic variants in collagen-related osteogenesis imperfecta". Molecular Genetics & Genomic Medicine. 7 (3): e559. doi:10.1002/mgg3.559. PMC 6418448. PMID 30675999.
- ^ Jump up to: a b c d e Van Dijk FS, Cobben JM, Kariminejad A, Maugeri A, Nikkels PG, van Rijn RR, Pals G (December 2011). "Osteogenesis Imperfecta: A Review with Clinical Examples". Molecular Syndromology. 2 (1): 1–20. doi:10.1159/000332228. PMC 3343766. PMID 22570641.
- ^ Subramanian S, Viswanathan VK (2021). Osteogenesis Imperfecta. StatPearls Publishing. StatPearls. PMID 30725642. Last Update: February 3, 2019.
- ^ Jump up to: a b c d Steiner RD, Pepin MG, Byers PH, Pagon RA, Bird TD, Dolan CR, Stephens K, Adam MP (6 May 2021). "COL1A1/2 Osteogenesis Imperfecta". In Adam MP, Ardinger HH, Pagon RA, et al. (eds.). GeneReviews [Internet]. University of Washington, Seattle. PMID 20301472. Archived from the original on 18 January 2017. Retrieved 26 August 2021.
- ^ Jump up to: a b Shapiro JR, Lietman C, Grover M, Lu JT, Nagamani SC, Dawson BC, et al. (July 2013). "Phenotypic variability of osteogenesis imperfecta type V caused by an IFITM5 mutation". Journal of Bone and Mineral Research. 28 (7): 1523–1530. doi:10.1002/jbmr.1891. PMC 3688672. PMID 23408678.
- ^ Jump up to: a b Moosa S, Yamamoto GL, Garbes L, Keupp K, Beleza-Meireles A, Moreno CA, et al. (October 2019). "Autosomal-Recessive Mutations in MESD Cause Osteogenesis Imperfecta". The American Journal of Human Genetics. 105 (4): 836–843. doi:10.1016/j.ajhg.2019.08.008. PMC 6817720. PMID 31564437.
- ^ Jump up to: a b c d e f g h i Van Dijk FS, Sillence DO (2014). "Osteogenesis imperfecta: Clinical diagnosis, nomenclature and severity assessment". American Journal of Medical Genetics Part A. 164 (6): 1470–1481. doi:10.1002/ajmg.a.36545. ISSN 1552-4833. PMC 4314691. PMID 24715559.
- ^ Jump up to: a b Singer RB, Ogston SA, Paterson CR (2001). "Mortality in various types of osteogenesis imperfecta". Journal of Insurance Medicine. 33 (3): 216–220. PMID 11558400.
- ^ "Osteogenesis Imperfecta: Types, Symptoms & Management". Cleveland Clinic. 5 May 2021. Retrieved 17 August 2021.
- ^ Jump up to: a b Basel D, Steiner RD (June 2009). "Osteogenesis imperfecta: recent findings shed new light on this once well-understood condition" (PDF). Genetics in Medicine. 11 (6): 375–385. doi:10.1097/GIM.0b013e3181a1ff7b. PMID 19533842.
- ^ Nadar R, Saraff V, Hogler W, Desai M, Shaw N (31 July 2019). "Determinants of survival in osteogenesis imperfecta (OI) Type II". Bone Abstracts. 7: 31. doi:10.1530/boneabs.7.P31. ISSN 2052-1219.
- ^ Zisa D, Shabbir A, Mastri M, Suzuki G, Lee T (1 November 2009). "Intramuscular VEGF repairs the failing heart: role of host-derived growth factors and mobilization of progenitor cells". American Journal of Physiology. 297 (5): 15809–15812. doi:10.1152/ajpregu.00227.2009. ISSN 0363-6119. PMC 2777764. PMID 19759338.
- ^ Georgescu I, Vlad C, Gavriliu TȘ, Dan S, Pârvan AA (15 June 2013). "Surgical treatment in Osteogenesis Imperfecta – 10 years experience". Journal of Medicine and Life. 6 (2): 205–213. ISSN 1844-122X. PMC 3725451. PMID 23904885.
- ^ Chen H (2006). "Triangular face". Atlas of genetic diagnosis and counseling. Totowa, NJ: Humana Press. p. 771. ISBN 978-1-58829-681-8. Archived from the original on 8 June 2013.
- ^ Lindahl K, Åström E, Rubin CJ, Grigelioniene G, Malmgren B, Ljunggren Ö, et al. (August 2015). "Genetic epidemiology, prevalence, and genotype–phenotype correlations in the Swedish population with osteogenesis imperfecta". European Journal of Human Genetics. 23 (8): 1042–1050. doi:10.1038/ejhg.2015.81. ISSN 1476-5438. PMC 4795106.
- ^ Glorieux FH, Rauch F, Plotkin H, Ward L, Travers R, Roughley P, et al. (September 2000). "Type V osteogenesis imperfecta: a new form of brittle bone disease". Journal of Bone and Mineral Research. 15 (9): 1650–1658. doi:10.1359/jbmr.2000.15.9.1650. PMID 10976985. S2CID 13748803.
- ^ Jump up to: a b c Cho TJ, Lee KE, Lee SK, Song SJ, Kim KJ, Jeon D, et al. (August 2012). Seoul National University Hospital. "A single recurrent mutation in the 5′-UTR of IFITM5 causes osteogenesis imperfecta type V". American Journal of Human Genetics. 91 (2): 343–348. doi:10.1016/j.ajhg.2012.06.005. PMC 3415533. PMID 22863190.
- ^ Jump up to: a b Morello R, Bertin TK, Chen Y, Hicks J, Tonachini L, Monticone M, et al. (20 October 2006). "CRTAP Is Required for Prolyl 3-Hydroxylation and Mutations Cause Recessive Osteogenesis Imperfecta". Cell. 127 (2): 291–304. doi:10.1016/j.cell.2006.08.039. ISSN 0092-8674.
- ^ Barnes AM, Chang W, Morello R, Cabral WA, Weis M, Eyre DR, et al. (28 December 2006). "Deficiency of Cartilage-Associated Protein in Recessive Lethal Osteogenesis Imperfecta". New England Journal of Medicine. 355 (26): 2757–2764. doi:10.1056/NEJMoa063804. ISSN 0028-4793. PMC 7509984. PMID 17192541.
- ^ Cabral WA, Chang W, Barnes AM, Weis M, Scott MA, Leikin S, et al. (March 2007). "Prolyl 3-hydroxylase 1 deficiency causes a recessive metabolic bone disorder resembling lethal/severe osteogenesis imperfecta". Nature Genetics. 39 (3): 359–365. doi:10.1038/ng1968. ISSN 1546-1718. PMC 7510175. PMID 17277775.
- ^ Van Dijk FS, Nesbitt IM, Zwikstra EH, Nikkels PG, Piersma SR, Fratantoni SA, et al. (October 2009). "PPIB Mutations Cause Severe Osteogenesis Imperfecta". The American Journal of Human Genetics. 85 (4): 521–527. doi:10.1016/j.ajhg.2009.09.001. ISSN 0002-9297. PMC 2756556. PMID 19781681.
- ^ Christiansen HE, Schwarze U, Pyott SM, AlSwaid A, Al Balwi M, Alrasheed S, et al. (March 2010). "Homozygosity for a Missense Mutation in SERPINH1, which Encodes the Collagen Chaperone Protein HSP47, Results in Severe Recessive Osteogenesis Imperfecta". The American Journal of Human Genetics. 86 (3): 389–398. doi:10.1016/j.ajhg.2010.01.034. ISSN 0002-9297. PMC 2833387. PMID 20188343.
- ^ Alanay Y, Avaygan H, Camacho N, Utine GE, Boduroglu K, Aktas D, et al. (April 2010). "Mutations in the Gene Encoding the RER Protein FKBP65 Cause Autosomal-Recessive Osteogenesis Imperfecta". The American Journal of Human Genetics. 86 (4): 551–559. doi:10.1016/j.ajhg.2010.02.022. ISSN 0002-9297. PMC 2850430. PMID 20362275.
- ^ Sam JE, Dharmalingam M (2017). "Osteogenesis Imperfecta". Indian Journal of Endocrinology and Metabolism. 21 (6): 903–908. doi:10.4103/ijem.IJEM_220_17. PMC 5729682. PMID 29285457.
- ^ Shapiro JR (2014). "Clinical and Genetic Classification of Osteogenesis Imperfecta and Epidemiology". Osteogenesis Imperfecta. Elsevier. pp. 15–22. doi:10.1016/b978-0-12-397165-4.00002-2. ISBN 9780123971654.
- ^ Jump up to: a b Sam JE, Dharmalingam M (1 November 2017). "Osteogenesis Imperfecta". Indian Journal of Endocrinology and Metabolism. 21 (6): 903–908. doi:10.4103/ijem.IJEM_220_17. PMC 5729682. PMID 29285457.
- ^ Shaheen R, Alazami AM, Alshammari MJ, Faqeih E, Alhashmi N, Mousa N, et al. (October 2012). "Study of autosomal recessive osteogenesis imperfecta in Arabia reveals a novel locus defined by TMEM38B mutation". Journal of Medical Genetics. 49 (10): 630–635. doi:10.1136/jmedgenet-2012-101142. ISSN 0022-2593.
- ^ Jump up to: a b Lindahl K, Åström E, Dragomir A, Symoens S, Coucke P, Larsson S, et al. (September 2018). "Homozygosity for CREB3L1 premature stop codon in first case of recessive osteogenesis imperfecta associated with OASIS-deficiency to survive infancy". Bone. 114: 268–277. doi:10.1016/j.bone.2018.06.019. ISSN 8756-3282.
- ^ Keller RB, Tran TT, Pyott SM, Pepin MG, Savarirayan R, McGillivray G, et al. (April 2018). "Monoallelic and biallelic CREB3L1 variant causes mild and severe osteogenesis imperfecta, respectively". Genetics in Medicine. 20 (4): 411–419. doi:10.1038/gim.2017.115. ISSN 1530-0366.
- ^ Mendoza-Londono R, Fahiminiya S, Majewski J, Tétreault M, Nadaf J, Kannu P, et al. (June 2015). "Recessive Osteogenesis Imperfecta Caused by Missense Mutations in SPARC". The American Journal of Human Genetics. 96 (6): 979–985. doi:10.1016/j.ajhg.2015.04.021. PMC 4457955. PMID 26027498.
- ^ Doyard M, Bacrot S, Huber C, Rocco MD, Goldenberg A, Aglan MS, et al. (1 April 2018). "FAM46A mutations are responsible for autosomal recessive osteogenesis imperfecta". Journal of Medical Genetics. The BMJ. 55 (4): 278–284. doi:10.1136/jmedgenet-2017-104999. ISSN 0022-2593. PMID 29358272.
- ^ Jump up to: a b Lindert U, Cabral WA, Ausavarat S, Tongkobpetch S, Ludin K, Barnes AM, et al. (6 July 2016). "MBTPS2 mutations cause defective regulated intramembrane proteolysis in X-linked osteogenesis imperfecta". Nature Communications. 7 (1): 11920. doi:10.1038/ncomms11920. ISSN 2041-1723. PMC 4935805. PMID 27380894.
- ^ Duy BH, Zhytnik L, Maasalu K, Kändla I, Prans E, Reimann E, et al. (1 December 2016). "Mutation analysis of the COL1A1 and COL1A2 genes in Vietnamese patients with osteogenesis imperfecta". Human Genomics. 10 (1): 27. doi:10.1186/s40246-016-0083-1. ISSN 1479-7364. PMC 4983065. PMID 27519266.
- ^ Palomo T, Vilaça T, Lazaretti-Castro M (December 2017). "Osteogenesis imperfecta: diagnosis and treatment". Current Opinion in Endocrinology, Diabetes and Obesity. 24 (6): 381–388. doi:10.1097/MED.0000000000000367. PMID 28863000. S2CID 4555427.
- ^ Valadares ER, Carneiro TB, Santos PM, Oliveira AC, Zabel B (18 July 2014). "What is new in genetics and osteogenesis imperfecta classification?". Jornal de Pediatria. 90 (6): 536–541. doi:10.1016/j.jped.2014.05.003. PMID 25046257.
- ^ Jump up to: a b c Forlino A, Cabral WA, Barnes AM, Marini JC (June 2011). "New perspectives on osteogenesis imperfecta". Nature Reviews. Endocrinology. 7 (9): 540–557. doi:10.1038/nrendo.2011.81. PMC 3443407. PMID 21670757.
- ^ Jump up to: a b Forlino A, Marini JC (April 2016). "Osteogenesis imperfecta". Lancet. 387 (10028): 1657–1671. doi:10.1016/S0140-6736(15)00728-X. PMC 7384887. PMID 26542481.
- ^ Koromani F, Trajanoska K, Rivadeneira F, Oei L (4 June 2019). "Recent Advances in the Genetics of Fractures in Osteoporosis". Frontiers in Endocrinology. doi:10.3389/fendo.2019.00337. ISSN 1664-2392. PMC 6559287. PMID 31231309.
- ^ Drögemüller C, Becker D, Brunner A, Haase B, Kircher P, Seeliger F, et al. (July 2009). Barsh GS (ed.). "A missense mutation in the SERPINH1 gene in Dachshunds with osteogenesis imperfecta". PLOS Genetics. 5 (7): e1000579. doi:10.1371/journal.pgen.1000579. PMC 2708911. PMID 19629171.
- ^ Rohrbach M, Giunta C (August 2012). "Recessive osteogenesis imperfecta: clinical, radiological, and molecular findings". American Journal of Medical Genetics. Part C, Seminars in Medical Genetics. 160C (3): 175–189. doi:10.1002/ajmg.c.31334. PMID 22791419. S2CID 28592112.
- ^ Jump up to: a b Marini JC, Reich A, Smith SM (August 2014). "Osteogenesis imperfecta due to mutations in non-collagenous genes: lessons in the biology of bone formation". Current Opinion in Pediatrics. 26 (4): 500–507. doi:10.1097/MOP.0000000000000117. PMC 4183132. PMID 25007323.
- ^ Hanagata N (March 2016). "IFITM5 mutations and osteogenesis imperfecta". Journal of Bone and Mineral Metabolism. 34 (2): 123–131. doi:10.1007/s00774-015-0667-1. PMID 26031935. S2CID 3173191.
- ^ Jump up to: a b c Pyott SM, Pepin MG, Schwarze U, Yang K, Smith G, Byers PH (February 2011). "Recurrence of perinatal lethal osteogenesis imperfecta in sibships: Parsing the risk between parental mosaicism for dominant mutations and autosomal recessive inheritance". Genetics in Medicine. 13 (2): 125–130. doi:10.1097/GIM.0b013e318202e0f6. ISSN 1530-0366.
- ^ Rauch F, Glorieux FH (April 2004). "Osteogenesis imperfecta". Lancet. 363 (9418): 1377–1385. doi:10.1016/S0140-6736(04)16051-0. PMID 15110498. S2CID 24081895.
- ^ Jump up to: a b Gautieri A, Uzel S, Vesentini S, Redaelli A, Buehler MJ (August 2009). "Molecular and mesoscale mechanisms of osteogenesis imperfecta disease in collagen fibrils". Biophysical Journal. 97 (3): 857–865. Bibcode:2009BpJ....97..857G. doi:10.1016/j.bpj.2009.04.059. PMC 2718154. PMID 19651044.
- ^ "Osteogenesis Imperfecta Foundation: Fast Facts". Archived from the original on 28 June 2007. Retrieved 5 July 2007.
- ^ El-Sobky TA, Shawky RM, Sakr HM, Elsayed SM, Elsayed NS, Ragheb SG, Gamal R (15 November 2017). "A systematized approach to radiographic assessment of commonly seen genetic bone diseases in children: A pictorial review". J Musculoskelet Surg Res. 1 (2): 25. doi:10.4103/jmsr.jmsr_28_17. S2CID 79825711.
- ^ Jameson JR, Albert CI, Busse B, Smith PA, Harris GF (29 March 2013). "3D micron-scale imaging of the cortical bone canal network in human osteogenesis imperfecta (OI)". In Weaver JB, Molthen RC (eds.). Medical Imaging 2013: Biomedical Applications in Molecular, Structural, and Functional Imaging. 8672. International Society for Optics and Photonics. pp. 86721L. doi:10.1117/12.2007209. S2CID 13876569.
- ^ Westgren M, Götherström C (September 2015). "Stem cell transplantation before birth - a realistic option for treatment of osteogenesis imperfecta?". Prenatal Diagnosis. 35 (9): 827–832. doi:10.1002/pd.4611. PMID 25962526. S2CID 10640427.
- ^ Jump up to: a b c Pepin MG, Byers PH (December 2015). "What every clinical geneticist should know about testing for osteogenesis imperfecta in suspected child abuse cases". American Journal of Medical Genetics. Part C, Seminars in Medical Genetics. 169 (4): 307–313. doi:10.1002/ajmg.c.31459. PMID 26566591. S2CID 26045033.
- ^ Glorieux FH, Bishop NJ, Plotkin H, Chabot G, Lanoue G, Travers R (October 1998). "Cyclic administration of pamidronate in children with severe osteogenesis imperfecta". The New England Journal of Medicine. 339 (14): 947–952. doi:10.1056/NEJM199810013391402. PMID 9753709. S2CID 19316414.Free full text
- ^ DiMeglio LA, Peacock M (January 2006). "Two-year clinical trial of oral alendronate versus intravenous pamidronate in children with osteogenesis imperfecta". Journal of Bone and Mineral Research. 21 (1): 132–140. doi:10.1359/JBMR.051006. PMID 16355282. S2CID 12996685.
- ^ Bishop N, Adami S, Ahmed SF, Antón J, Arundel P, Burren CP, et al. (October 2013). "Risedronate in children with osteogenesis imperfecta: a randomised, double-blind, placebo-controlled trial". Lancet. 382 (9902): 1424–1432. doi:10.1016/S0140-6736(13)61091-0. PMID 23927913. S2CID 25559791.
- ^ Ward LM, Rauch F (October 2013). "Oral bisphosphonates for paediatric osteogenesis imperfecta?". Lancet. 382 (9902): 1388–1389. doi:10.1016/S0140-6736(13)61531-7. PMID 23927912. S2CID 5872511.
- ^ Rothschild L, Goeller JK, Voronov P, Barabanova A, Smith P (November 2018). "Anesthesia in children with osteogenesis imperfecta: Retrospective chart review of 83 patients and 205 anesthetics over 7 years". Pediatric Anesthesia. 28 (11): 1050–1058. doi:10.1111/pan.13504.
- ^ Houck JP (2014). "Osteogenesis imperfecta". In Houck JP, Hache M, Sun LS (eds.). Handbook of Pediatric Anesthesia. New York: McGraw-Hill Education. ISBN 9780071769358.
- ^ Goeller JK, Rothschild L (2020), Kruse RW (ed.), "Anesthetic and Post-operative Pain Management" (ePub), Osteogenesis Imperfecta: A Case-Based Guide to Surgical Decision-Making and Care, Springer International Publishing, pp. 111–125, doi:10.1007/978-3-030-42527-2_8, ISBN 978-3-030-42527-2
- ^ Esposito P, Wallace MJ (2020), Kruse RW (ed.), "Osteogenesis Imperfecta Surgical Management of the Femur and Knee" (ePub), Osteogenesis Imperfecta: A Case-Based Guide to Surgical Decision-Making and Care, Springer International Publishing, pp. 147–182, doi:10.1007/978-3-030-42527-2_10, ISBN 978-3-030-42527-2
- ^ "Financial Assistance". Shriners Hospitals for Children. Archived from the original on 3 March 2021. Retrieved 21 August 2021.
Shriners Hospitals for Children is committed to providing specialized care to children with orthopaedic conditions [...] regardless of the families' ability to pay.
- ^ "A Leader in the Treatment of Osteogensis Imperfecta (OI)". Shriners International. Archived from the original on 28 September 2007. Retrieved 5 July 2007.
- ^ Sofield HA, Millar EA (1 December 1959). "Fragmentation, Realignment, and Intramedullary Rod Fixation of Deformities of the Long Bones in Children: A Ten-Year Appraisal". The Journal of Bone and Joint Surgery. 41 (8): 1371–1391. doi:10.2106/00004623-195941080-00001. ISSN 0021-9355.
- ^ "The Fassier-Duval Telescopic IM System". Pega Medical. Laval, Quebec. Retrieved 15 August 2021.
- ^ Jump up to: a b Sterian A, Balanescu R, Barbilian A, Ulici A (2015). "Osteosynthesis in Osteogenesis Imperfecta, telescopic versus non-telescopic nailing". Journal of Medicine and Life. 8 (4): 563–565. PMC 4656972. PMID 26664490.
- ^ Jump up to: a b Frassier F, Duval P, Paley D (2021). "Fassier–Duval Telescopic IM System Surgical Technique" (PDF). Pega Medical. Laval, Quebec. FD-ST-EN Revision K. Retrieved 15 August 2021.
- ^ Belyea CM, Knox JB (1 January 2020). "Spinal fusion in children with osteogenesis imperfecta: A nationwide retrospective comparative cohort study over a 12-year period". Current Orthopaedic Practice. 31 (1): 72–75. doi:10.1097/BCO.0000000000000805. ISSN 1940-7041. S2CID 209229412.
- ^ Zani A, Ford-Adams M, Ratcliff M, Bevan D, Inge TH, Desai A (January 2017). "Weight loss surgery improves quality of life in pediatric patients with osteogenesis imperfecta". Surgery for Obesity and Related Diseases. Elsevier Inc. 13 (1): 41–44. doi:10.1016/j.soard.2015.11.029. PMC 5965274. PMID 26948942.
- ^ Semler O, Fricke O, Vezyroglou K, Stark C, Schoenau E (1 January 2007). "Preliminary results on the mobility after whole body vibration in immobilized children and adolescents". Journal of Musculoskeletal & Neuronal Interactions. 7 (1): 77–81. ISSN 1108-7161. PMID 17396011.
- ^ Jump up to: a b Biria M, Abbas FM, Mozaffar S, Ahmadi R (July 2012). "Dentinogenesis imperfecta associated with osteogenesis imperfecta". Dental Research Journal. 9 (4): 489–494. PMC 3491340. PMID 23162594.
- ^ Green, Rick (11 March 2012). "Brittle Bone Disease Forced Mother Into 'No Easy Choice'". Hartford Courant. Retrieved 16 September 2021.
- ^ Hopper, Tristin (6 February 2013). "'Incompatible with life': Mother's account of her difficult decision to request a late-term abortion sheds light on 'live-born' debate". National Post. Retrieved 16 September 2021.
- ^ Paterson CR, Ogston SA, Henry RM (10 February 1996). "Life expectancy in osteogenesis imperfecta". British Medical Journal. 312 (7027): 351. doi:10.1136/bmj.312.7027.351. ISSN 0959-8138. PMC 2350292. PMID 8611834.
- ^ Plotkin H (29 February 2016). "Genetics of Osteogenesis Imperfecta". Archived from the original on 30 December 2010.
- ^ "Osteogenesis Imperfecta Panel". University of Nebraska Medical Center. Retrieved 30 July 2019.
- ^ Abali S, Arman A, Atay Z, Bereket A, Bas S, Haliloglu B, et al. (19 August 2016). "Frequency of Recessive Osteogenesis Imperfecta in a Turkish Cohort and Genetic Causes". ESPE Abstracts. Bioscientifica. 86: 131.
- ^ Jump up to: a b c Roger S, Francis MJ, Houghton GR (1983). The brittle bone syndrome: osteogenesis imperfecta. London: Butterworth-Heinemann. p. 4. ISBN 978-0-407-00211-1. OCLC 9784850.
- ^ Lowenstein EJ (May 2009). "Osteogenesis imperfecta in a 3,000-year-old mummy". Child's Nervous System. 25 (5): 515–516. doi:10.1007/s00381-009-0817-7. ISSN 1433-0350.
- ^ Jump up to: a b c Tainmont J (2007). "History of osteogenesis imperfecta or brittle bone disease: a few stops on a road 3000 years long". B-ENT. 3 (3): 157–173. ISSN 1781-782X. PMID 17970442.
- ^ Groeneveld E (12 November 2018). "Ivar the Boneless". World History Encyclopedia. Ancient History Encyclopedia Limited. Archived from the original on 21 August 2021. Retrieved 21 August 2021.CS1 maint: uses authors parameter (link)
- ^ Lewis R, Lotha G, Gaur A (8 January 2019). "Ivar the Boneless". Encyclopedia Britannica. Retrieved 21 August 2021.CS1 maint: uses authors parameter (link)
- ^ Malebranche N (1688). (1842 ed.). p. 119 – via Wikisource.
- ^ Edelu BO, Ndu IK, Asinobi IN, Obu HA, Adimora GN (2014). "Osteogenesis Imperfecta: A Case Report and Review of Literature". Annals of Medical and Health Sciences Research. Enugu State: Nigerian Medical Association. 4 (Suppl 1): S1–S5. doi:10.4103/2141-9248.131683. ISSN 2141-9248. PMC 4083720. PMID 25031897.
- ^ Hektoen L (May 1903). "Anatomical Study of a Short-Limbed Dwarf, With Special Reference To Osteogenesis Imperfecta and Chondrodystrophia Foetalis". The American Journal of the Medical Sciences. J. B. Lippincott & Co. 125 (5): 751–769. doi:10.1097/00000441-190305000-00001. ISBN 978-0-243-38392-4. S2CID 71836609.
- ^ Adair-Dighton CA (1912). "Four generations of blue sclerotics". The Ophthalmoscope. London: George Pulman & Sons, Ltd. 10: 188–189. OCLC 1049881577 – via Internet Archive.
- ^ Vrolik W (1849). Tabulae ad illustrandam embryogenesin hominis et mammalium, tam naturalem quam abnormem [Illustrations of human and mammalian embryogenesis, both normal and abnormal] (in Latin and Dutch). Amsterdam: G.M.P. Londonck. pp. 562–566. OCLC 489920691.
- ^ Jump up to: a b Baljet B (January 2002). "Aspects of the history of osteogenesis imperfecta (Vrolik's syndrome)". Annals of Anatomy. 184 (1): 1–7. doi:10.1016/S0940-9602(02)80023-1. PMID 11876477.
- ^ Baljet B (2002). "Willem Vrolik and "his" Syndrome". In Rauschmann MA, Thomann KD, Zichner, L (eds.). Deutsches Orthopädisches Geschichts und Forschungsmuseum. 4. Heidelberg: Verlag Theodor Steinkopff. pp. 133–144. doi:10.1007/978-3-642-57510-5_15. ISBN 978-3-642-57510-5.
- ^ Looser E (1906). "Zur Kenntnis der Osteogenesis Imperfecta Congenita et Tarda (sogenannte idiopathische Osteopsatyrosis)" [On awareness of osteogenesis imperfecta congenita and tarda (so-called "idiopathic osteopsatyrosis")]. Mitteilungen aus den Grenzgebieten der Medizin und Chirurgie (Dispatches from the Frontier of Medicine and Surgery) (in German). 15: 161–207.
- ^ synd/1743 at Who Named It?
- ^ Rasmussen C, Danks A (2010). Double helix, double joy: David Danks, the father of clinical genetics in Australia. Carlton, Victoria: Melbourne University Press. p. 116. ISBN 978-0-522-85799-3. OCLC 906592657.
- ^ Jump up to: a b Warman ML, Cormier-Daire V, Hall C, Krakow D, Lachman R, LeMerrer M, et al. (May 2011). "Nosology and classification of genetic skeletal disorders: 2010 revision". American Journal of Medical Genetics Part A. 155 (5): 943–968. doi:10.1002/ajmg.a.33909. PMC 3166781. PMID 21438135.
- ^ Jump up to: a b "Osteogenesis Imperfecta (OI) in Dachshunds". UC Davis Veterinary Genetics Laboratory. UC Davis School of Veterinary Medicine. Retrieved 28 August 2021.
- ^ Jump up to: a b "Osteogenesis Imperfecta (OI Dachsund)". Center for Animal Genetics. Generatio GmbH. Archived from the original on 8 November 2018. Retrieved 7 November 2018.
- ^ Jump up to: a b c d Enderli TA, Burtch SR, Templet JN, Carriero A (September 2016). "Animal models of osteogenesis imperfecta: applications in clinical research". Orthopedic Research and Reviews. 8: 41–55. doi:10.2147/ORR.S85198. PMC 6209373. PMID 30774469.
- ^ Saban J, Zussman MA, Havey R, Patwardhan AG, Schneider GB, King D (December 1996). "Heterozygous oim mice exhibit a mild form of osteogenesis imperfecta". Bone. 19 (6): 575–579. doi:10.1016/S8756-3282(96)00305-5.
External links[]
| Wikimedia Commons has media related to Osteogenesis imperfecta. |
| Classification | |
|---|---|
| External resources |
|
- "Osteogenesis Imperfecta Overview". NIH Osteoporosis and Related Bone Diseases — National Resource Center. National Institutes of Health, U.S. Department of Health and Human Services.
- Abnormalities of dermal fibrous and elastic tissue
- Skeletal disorders
- Collagen disease
- Rare diseases