From Wikipedia, the free encyclopedia
For other uses, see Vitisin .
Vitisin C
Names
Preferred IUPAC name
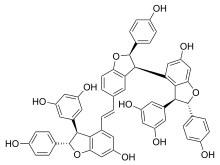
(22 S ,23 S ,32 R ,33 S ,4E ,62 S ,63 S )-22 ,32 ,62 -Tris(4-hydroxyphenyl)-22 ,23 ,32 ,33 ,62 ,63 -hexahydro-2(3,4),3(3,5),6(4,3)-tris([1]benzofurana)-1,7(1)-dibenzenaheptaphan-4-ene-13 ,15 ,26 ,66 ,73 ,75 -hexol
Identifiers
3D model (JSmol )
ChEMBL
ChemSpider
InChI=1S/C56H42O12/c57-35-10-4-29(5-11-35)54-50(33-19-38(60)23-39(61)20-33)49-32(18-42(64)26-47(49)67-54)3-1-28-2-16-46-44(17-28)52(56(66-46)31-8-14-37(59)15-9-31)45-25-43(65)27-48-53(45)51(34-21-40(62)24-41(63)22-34)55(68-48)30-6-12-36(58)13-7-30/h1-27,50-52,54-65H/b3-1+/t50-,51-,52+,54+,55+,56-/m0/s1
Y Key: WZKKRZSJTLGPHH-WHSOPTDBSA-N
Y InChI=1/C56H42O12/c57-35-10-4-29(5-11-35)54-50(33-19-38(60)23-39(61)20-33)49-32(18-42(64)26-47(49)67-54)3-1-28-2-16-46-44(17-28)52(56(66-46)31-8-14-37(59)15-9-31)45-25-43(65)27-48-53(45)51(34-21-40(62)24-41(63)22-34)55(68-48)30-6-12-36(58)13-7-30/h1-27,50-52,54-65H/b3-1+/t50-,51-,52+,54+,55+,56-/m0/s1
Key: WZKKRZSJTLGPHH-WHSOPTDBBK
OC(C=C1)=CC=C1[C@@H](O2)[C@@H](C3=CC(O)=CC(O)=C3)C4=C2C=C(O)C=C4/C=C/C5=CC([C@H](C6=C([C@H](C7=CC(O)=CC(O)=C7)[C@@H](C8=CC=C(O)C=C8)O9)C9=CC(O)=C6)[C@H](C%10=CC=C(O)C=C%10)O%11)=C%11C=C5
Oc1ccc(cc1)[C@H]4Oc2cc(O)cc(c2[C@@H]4c3cc(O)cc(O)c3)\C=C\c%10ccc%11O[C@@H](c5ccc(O)cc5)[C@@H](c7cc(O)cc8O[C@H](c6ccc(O)cc6)[C@H](c78)c9cc(O)cc(O)c9)c%11c%10
Properties
C 56 H 42 O 12
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
N what is Y N
Infobox references
Chemical compound
Vitisin C is a hydroxystilbenoid . It is a resveratrol tetramer found in plants of the genus Vitis [1] [2]
References [ ]
^ Ito, J (1996). "Absolute structures of new hydroxystilbenoids, vitisin C and viniferal, from Vitis vinifera 'Kyohou' ". Tetrahedron . 52 (30): 9991–9998. doi :10.1016/0040-4020(96)00543-1 . ^ Seya, K; Furukawa, K; Taniguchi, S; Kodzuka, G; Oshima, Y; Niwa, M; Motomura, S (2003). "Endothelium-dependent vasodilatory effect of vitisin C, a novel plant oligostilbene from Vitis plants (Vitaceae), in rabbit aorta" . Clinical Science . 105 (1): 73–9. doi :10.1042/CS20020288 . PMID 12605596 . S2CID 41885739 .
External links [ ]
Dimers Trimers Tetramers:
(3"-hydroxygnetin E)
(3"-methoxygnetin E)
(isorhapontigenin tetramer)
and Higher polymers Oligomeric forms
Dimers Trimers Tetramers Pentamers Hexamers Higher polymers
Glycosides or conjugates
Diptoindonesin A (C-glucoside of ε-viniferin)Foeniculoside I (glucoside of miyabenol C), , and (an ε-viniferin-ascorbic acid hybrid compound)
(O-glucoside of ampelopsin A)
Categories :
Resveratrol oligomers Natural phenol tetramers Grape Aromatic compound stubs Hidden categories:
Chemical articles with multiple compound IDs Multiple chemicals in an infobox that need indexing Articles without KEGG source Articles without UNII source Chembox CAS registry number linked Articles with changed CASNo identifier Pages using collapsible list with both background and text-align in titlestyle Articles containing unverified chemical infoboxes Articles with short description Short description matches Wikidata All stub articles