Lenacapavir
 | |
| Clinical data | |
|---|---|
| Other names | GS-6207 |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| Chemical and physical data | |
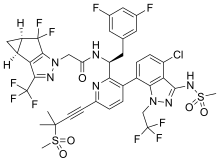
| Formula | C41H36ClF8N7O5S2 |
| Molar mass | 958.34 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lenacapavir (development code GS-6207) is an experimental drug for the treatment of HIV being developed by Gilead Sciences.[1] As of 2021, it is in Phase 2/3 clinical trials.[2] It is being investigated as a treatment for HIV patients infected with multidrug-resistant virus and as a twice-yearly injectable for pre-exposure prophylaxis (PrEP).[2][3]
If successful in clinical trials, it would become a first-in-class HIV capsid inhibitor for the treatment of HIV.[4]
References[]
- ^ Link JO, Rhee MS, Tse WC, Zheng J, Somoza JR, Rowe W, et al. (August 2020). "Clinical targeting of HIV capsid protein with a long-acting small molecule". Nature. 584 (7822): 614–618. Bibcode:2020Natur.584..614L. doi:10.1038/s41586-020-2443-1. PMC 8188729. PMID 32612233. S2CID 220293679.
- ^ a b Boerner H (11 March 2021). "Lenacapavir Effective in Multidrug Resistant HIV". Medscape.
- ^ Highleyman L (15 March 2021). "Lenacapavir Shows Promise for Long-Acting HIV Treatment and Prevention". POZ.
- ^ Gallagher GM (12 March 2020). "Trial Data Released for Investigational HIV-1 Capsid Inhibitor". Contagion Live.
Categories:
- Antiretroviral drugs
- Cyclopropanes
- Indazoles
- Trifluoromethyl compounds
- Chloroarenes
- Fluoroarenes
- Sulfonamides
- Pyridines
- Amides
- Antiinfective agent stubs