Menadione
This article is missing information about drugbox or more Chembox Pharmacology. (January 2021) |

| |
| |
| Names | |
|---|---|
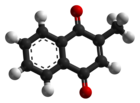
| Preferred IUPAC name
2-Methylnaphthalene-1,4-dione | |
| Other names
Menaphthone; Vitamin K3; β-Methyl-1,4-naphthoquinone; 2-Methyl-1,4-naphthodione; 2-Methyl-1,4-naphthoquinone
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.338 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| C11H8O2 | |
| Molar mass | 172.183 g·mol−1 |
| Appearance | Bright yellow crystals |
| Density | 1.225g/cm3 |
| Melting point | 105 to 107 °C (221 to 225 °F; 378 to 380 K) |
| Insoluble | |
| Pharmacology | |
| B02BA02 (WHO) | |
| |
| |
| Legal status |
|
| Hazards | |
| Flash point | 113.8 °C (236.8 °F; 386.9 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
0.5 g/kg (oral, mouse) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Menadione is a natural[3] organic compound with the formula C6H4(CO)2C2H(CH3). It is an analog of 1,4-naphthoquinone with a methyl group in the 2-position.[4] It is sometimes called vitamin K3. Use is allowed as a nutritional supplement in animal feed because of its vitamin K activity.
Biochemistry[]
Menadione is converted to vitamin K2 (specifically, MK-4) by the prenyltransferase action of vertebrate UBIAD1.[3] This reaction requires the hydroquinone (reduced) form of K3, menadiol, produced by an unidentified enzyme.[5]
Menadione is also a circulating form of vitamin K, produced in small amounts (1–5%) after intestinal absorption of K1 and K2. This circulation explains the uneven tissue distribution of MK-4, especially since menadione can penetrate the blood-brain barrier. The cleavage enzyme is yet to be identified.[5]
Terminology[]
The compound is variously known as vitamin K3[6] and provitamin K3.[7] Proponents of the latter name generally argue that the compound is not a real vitamin due to its artificial status (prior to its identification as a circulating intermediate) and its lack of a 3-methyl side chain preventing it from exert all the functions[which?] of the K vitamins.
Uses[]

It is an intermediate in the chemical synthesis of vitamin K by first reduction to the diol menadiol, which is susceptible to coupling to the phytol.[8]
Despite the fact that it can serve as a precursor to various types of vitamin K, menadione is generally not used as a nutritional supplement in economically developed countries. Menadione for human use at pharmaceutical strength is available in some countries with large lower income populations, such as India.[2] The typical daily dose is 10 mg oral or 2 mg parenteral.[9] It is used in the treatment of hypoprothrombinemia outside of the United States.[2]
Toxicity[]
This section is missing information about mechanism (redox?). (January 2021) |
Large doses[clarification needed] of menadione have been reported to cause adverse outcomes including hemolytic anemia due to glucose-6-phosphate dehydrogenase deficiency, neonatal brain or liver damage, or neonatal death in some rare cases. In the United States, over-the-counter menadione supplements are banned by the U.S. Food and Drug Administration because of their potential toxicity in human use.[citation needed]
Low-dose menadione is still used as an inexpensive micronutrient for livestock in many countries. Forms of menadione are also included in some pet foods in developed countries as a source of vitamin K. These doses have yielded no reported cases of toxicity from menadione in livestock or pets. Although handling may be hazardous, the European Food Safety Authority found in 2013 that it is an effective source of vitamin K in animal nutrition that does not pose a risk to the environment.[10]
References[]
- ^ The Merck Index, 11th Edition, 5714
- ^ Jump up to: a b c "Menadione drug information". DrugsUpdate India.
- ^ Jump up to: a b Hirota, Yoshihisa; Tsugawa, Naoko; Nakagawa, Kimie; Suhara, Yoshitomo; Tanaka, Kiyoshi; Uchino, Yuri; Takeuchi, Atsuko; Sawada, Natsumi; Kamao, Maya; Wada, Akimori; Okitsu, Takashi (2013-11-15). "Menadione (vitamin K3) is a catabolic product of oral phylloquinone (vitamin K1) in the intestine and a circulating precursor of tissue menaquinone-4 (vitamin K2) in rats". The Journal of Biological Chemistry. 288 (46): 33071–33080. doi:10.1074/jbc.M113.477356. ISSN 1083-351X. PMC 3829156. PMID 24085302.
- ^ Castro FA, Mariani D, Panek AD, Eleutherio EC, Pereira MD (2008). Fox (ed.). "Cytotoxicity mechanism of two naphthoquinones (menadione and plumbagin) in Saccharomyces cerevisiae". PLOS ONE. 3 (12): e3999. Bibcode:2008PLoSO...3.3999C. doi:10.1371/journal.pone.0003999. PMC 2600608. PMID 19098979.
- ^ Jump up to: a b Shearer, Martin J.; Newman, Paul (March 2014). "Recent trends in the metabolism and cell biology of vitamin K with special reference to vitamin K cycling and MK-4 biosynthesis". Journal of Lipid Research. 55 (3): 345–362. doi:10.1194/jlr.R045559. ISSN 0022-2275. PMC 3934721. PMID 24489112.
- ^ Scott GK, Atsriku C, Kaminker P, Held J, Gibson B, Baldwin MA, Benz CC (September 2005). "Vitamin K3 (menadione)-induced oncosis associated with keratin 8 phosphorylation and histone H3 arylation". Molecular Pharmacology. 68 (3): 606–15. doi:10.1124/mol.105.013474. PMID 15939799. S2CID 19076885.
- ^ "Vitamin K". Linus Pauling Institute. 2014-04-22. Retrieved 2021-01-28.
- ^ Weber F, Rüttimann A (2012). "Vitamin K". Ullmann's Encyclopedia Of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.o27_o08.
- ^ "Menadione (B02BA02)". WHOCC - ATC/DDD Index.
- ^ EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP) (January 2014). "Scientific Opinion on the safety and efficacy of vitamin K3 (menadione sodium bisulphite and menadione nicotinamide bisulphite) as a feed additive for all animal species". EFSA Journal. 12 (1): 3532. doi:10.2903/j.efsa.2014.3532.
External links[]
- Menadione in the Pesticide Properties DataBase (PPDB)
- 1,4-Naphthoquinones
- Vitamin K
