Lead(II) acetate

| |

| |
| Names | |
|---|---|
| IUPAC name
Lead(II) acetate
| |
| Systematic IUPAC name
Lead(II) ethanoate | |
| Other names
Plumbous acetate, Salt of Saturn, Sugar of Lead, Lead diacetate
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.551 |
| EC Number |
|
| MeSH | lead+acetate |
PubChem CID
|
|
| RTECS number |
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| Pb(C2H3O2)2 | |
| Molar mass | 325.29 g/mol (anhydrous) 379.33g/mol (trihydrate) |
| Appearance | White powder or colourless, efflorescent crystals |
| Odor | Slightly acetic |
| Density | 3.25 g/cm3 (20 °C, anhydrous) 2.55 g/cm3 (trihydrate) 1.69 g/cm3 (decahydrate)[1] |
| Melting point | 280 °C (536 °F; 553 K) (anhydrous) 75 °C (167 °F; 348 K) (trihydrate) decomposes[4] at ≥ 200 °C 22 °C (72 °F; 295 K) (decahydrate)[1] |
| Boiling point | Decomposes |
| Anhydrous: 19.8 g/100 mL (0 °C) 44.31 g/100 mL (20 °C) 69.5 g/100 mL (30 °C)[2] 218.3 g/100 mL (50 °C)[1] | |
| Solubility | Anhydrous and trihydrate are soluble in alcohol, glycerol[2] |
| Solubility in methanol | Anhydrous:[2] 102.75 g/100 g (66.1 °C) Trihydrate:[3] 74.75 g/100 g (15 °C) 214.95 g/100 g (66.1 °C) |
| Solubility in glycerol | Anhydrous:[2] 20 g/100 g (15 °C) Trihydrate:[3] 143 g/100 g (20 °C) |
| −89.1·10−6 cm3/mol | |
Refractive index (nD)
|
1.567 (trihydrate)[1] |
| Structure | |
| Monoclinic (anhydrous, trihydrate) Rhombic (decahydrate) | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−960.9 kJ/mol (anhydrous)[2] −1848.6 kJ/mol (trihydrate)[3] |
| Hazards | |
| Main hazards | Neurotoxic, probable human carcinogen |
| GHS pictograms |   [4] [4]
|
| GHS Signal word | Danger |
GHS hazard statements
|
H360, H373, H410[4] |
| P201, P273, P308+313, P501[4] | |
| NFPA 704 (fire diamond) | 
2
1
1 |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
400 mg/kg (mice, oral)[1] |
LCLo (lowest published)
|
300 mg/kg (dog, oral)[5] |
| Related compounds | |
Other cations
|
Lead(IV) acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Lead(II) acetate (Pb(CH3COO)2), also known as lead acetate, lead diacetate, plumbous acetate, sugar of lead, lead sugar, salt of Saturn, or Goulard's powder, is a white crystalline chemical compound with a slightly sweet taste. Like many other lead compounds, it is toxic. Lead acetate is soluble in water and glycerin. With water it forms the trihydrate, Pb(CH3COO)2·3H2O, a colourless or white efflorescent monoclinic crystalline substance.
The substance is used as a reagent to make other lead compounds and as a fixative for some dyes. In low concentrations, it is the principal active ingredient in progressive types of hair colouring dyes.[6] Lead(II) acetate is also used as a mordant in textile printing and dyeing, and as a drier in paints and varnishes. It was historically used as a sweetener and preservative in wines and in other foods and for cosmetics.
Production[]
Lead acetate can be made by boiling elemental lead in acetic acid and hydrogen peroxide. This method will also work with lead carbonate or lead oxide.
Pb(s) + H2O2(aq) + 2 H+(aq) → Pb2+(aq) + 2 H2O(l)
Pb2+(aq) + 2 CH3COO−(aq) → Pb(CH3COO)2(aq)
Lead(II) acetate can also be made via a single displacement reaction between copper acetate and lead metal:
Cu(CH3COO)2 + Pb → Cu + Pb(CH3COO)2
Structure[]
The crystal structure of anhydrous lead(II) acetate has been described as a 2D coordination polymer. In comparison, lead(II) acetate trihydrate's structure is a 1D coordination polymer.[7] In the trihydrate, the Pb2+ ion's coordination sphere consists of nine oxygen atoms belonging to three water molecules, two bidentate acetate groups and two bridging acetate groups. The coordination geometry at Pb is a monocapped square antiprism.[8][9] The trihydrate thermally decomposes to a hemihydrate, Pb(OAc)2·½H2O, and to basic acetates such as Pb4O(OAc)6 and Pb2O(OAc)2.[7]
| Degree of hydration | Pb coordination | Strongly bonded aggregation | Weakly bonded aggregation |
|---|---|---|---|
| Anhydrous[7] Pb(OAc)2 |

|
 2D sheet |
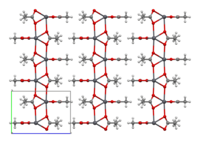 sheets stacked with hydrophobic surfaces in contact |
| Trihydrate[8][9] Pb(OAc)2·3H2O |

|
 1D chain |
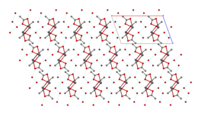 chains linked by hydrogen bonds |
Uses[]
Sweetener[]
Like other lead(II) salts, lead(II) acetate has a sweet taste, which led to its historical use as a sugar substitute in both wines and foods.[10] The ancient Romans, who had few sweeteners besides honey, would boil must (grape juice) in lead pots to produce a reduced sugar syrup called defrutum, concentrated again into sapa. This syrup was used to sweeten wine and to sweeten and preserve fruit. It is possible that lead(II) acetate or other lead compounds leaching into the syrup might have caused lead poisoning in those who consumed it.[11] Lead acetate is no longer used in the production of sweeteners because of its recognized toxicity. Modern chemistry can easily detect it, which has almost completely stopped the illegal use that continued decades after its use as a sweetener was banned.[12]
Historical incidents[]
The earliest confirmed poisoning by lead acetate was that of Pope Clement II who died in October 1047. A toxicological examination of his remains conducted in the mid-20th century confirmed centuries-old rumors that he had been poisoned with lead sugar.[13] It is not clear whether he was assassinated.
In 1787 painter Albert Christoph Dies swallowed, by accident, approximately 3/4 oz (20 g) of lead acetate. His recovery from this poison was slow and incomplete. He lived with illnesses until his death in 1822.[14][15]
Although the use of lead(II) acetate as a sweetener was already illegal at that time, composer Ludwig van Beethoven may have died of lead poisoning caused by wines adulterated with lead acetate (see also Beethoven's liver).[16][17]
In the 1850s, Mary Seacole applied lead(II) acetate, among other remedies, against an epidemic of cholera in Panama.[18][19]
In 1887, 38 hunting horses belonging to Captain William Hollwey Steeds were poisoned in their stables at Clonsilla House, Dublin, Ireland. At least ten of the hunters died. Captain Steeds, an 'extensive commission agent', had previously supplied the horses for the Bray and Greystones Coach. It transpired they had been fed a bran mash that had been sweetened with a toxic lead acetate.[20]
Cosmetics[]
Lead(II) acetate, as well as white lead, has been used in cosmetics throughout history.[21]
Until recently, it was still used in the USA in men's hair colouring products[22] like Grecian Formula. It was not until just a few years ago when the manufacturer removed lead acetate from the hair coloring product, and as of July 2018 the ingredients in Grecian Formula are water, isopropyl alcohol, triethanolamine, bismuth citrate, sodium thiosulfate, fragrance, and panthenol. Lead acetate has been replaced by bismuth citrate as the progressive colorant. Its use in cosmetics has been banned in Canada by Health Canada in 2005 (effective at the end of 2006) based on tests showing possible carcinogenicity and reproductive toxicity,[23] and it is also banned in the European Union[23] and has been on the California Proposition 65 warning list as a carcinogen since 1988.[24]
Medical uses[]
Lead(II) acetate solution was a commonly used folk remedy for sore nipples.[25] In modern medicine, for a time, it was used as an astringent, in the form of Goulard's Extract, and it has also been used to treat poison ivy.[26]
Industrial uses[]
Lead(II) acetate paper is used to detect the poisonous gas hydrogen sulfide. The gas reacts with lead(II) acetate on the moistened test paper to form a grey precipitate of lead(II) sulfide.
An aqueous solution of lead(II) acetate is the byproduct of a 1:1 ratio of hydrogen peroxide and white vinegar (acetic acid) used in the cleaning and maintenance of stainless steel firearm suppressors (silencers) and compensators. The solution is agitated by the bubbling action of the hydrogen peroxide, and the main reaction is the dissolution of lead deposits within the suppressor by the acetic acid, which forms lead acetate. Because of its high toxicity, this chemical solution must be appropriately disposed by a chemical processing facility or hazardous materials centre. Alternatively, the solution may be reacted with sulfuric acid to precipitate nearly insoluble lead(II) sulfate. The solid may then be removed by mechanical filtration and is safer to dispose of than aqueous lead acetate.
It was also used in making of slow matches during the Middle Ages. It was made by mixing natural form of lead(II) oxide called litharge and vinegar.
Sugar of lead was a recommended agent added to linseed oil during heating to produce "boiled" linseed oil, the lead and heat acting to cause the oil to cure faster than raw linseed oil.[27]
See also[]
References[]
- ^ Jump up to: a b c d e Pradyot, Patnaik (2003). Handbook of Inorganic Chemicals. The McGraw-Hill Companies, Inc. ISBN 0-07-049439-8.
- ^ Jump up to: a b c d e http://chemister.ru/Database/properties-en.php?dbid=1&id=1990
- ^ Jump up to: a b c http://chemister.ru/Database/properties-en.php?dbid=1&id=1762
- ^ Jump up to: a b c d Sigma-Aldrich Co., Lead(II) acetate trihydrate. Retrieved on 2014-06-08.
- ^ "Lead compounds (as Pb)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "Lead Acetate in 'Progressive' Hair Dye Products". fda.gov.
- ^ Jump up to: a b c Martínez-Casado, Francisco J.; Ramos-Riesco, Miguel; Rodríguez-Cheda, José A.; Cucinotta, Fabio; Matesanz, Emilio; Miletto, Ivana; Gianotti, Enrica; Marchese, Leonardo; Matěj, Zdeněk (2016). "Unraveling the Decomposition Process of Lead(II) Acetate: Anhydrous Polymorphs, Hydrates, and Byproducts and Room Temperature Phosphorescence". Inorg. Chem. 55 (17): 8576–8585. doi:10.1021/acs.inorgchem.6b01116.
- ^ Jump up to: a b Rajaram, R. K.; Mohana Rao, J. K. (1982). "Crystal structure of lead acetate trihydrate". Z. Kristallogr. 160 (1–4): 225–233. doi:10.1524/zkri.1982.160.14.225.
- ^ Jump up to: a b Bryant, Robert G.; Chacko, V. P.; Etter, Margaret C. (1984). "Carbon-13 CP/MAS NMR and crystallographic investigations of the structure and solid-state transformations of lead(II) acetate trihydrate". Inorg. Chem. 23 (22): 3580–3584. doi:10.1021/ic00190a029.
- ^ "The Disturbingly Long History of Lead Toxicity in Winemaking." Anna Archibald, 30 July 2020. Retrieved: 22 December, 2020.
- ^ Lead Poisoning and Rome
- ^ Stoeppler, M. (1992), Hazardous Metals in the Environment, Techniques and Instrumentation in Analytical Chemistry, 12, Elsevier, p. 60, ISBN 9780080875606,
From the results achieved so far it is obvious that the purity law for lead in wines in the last two centuries was frequently ignored.
- ^ Specht W and Fischer K (1959). Vergiftungsnachweis an den Resten einer 900 Jahre alten Leiche. Arch. Kriminol., 124: 61-84. [Translation:Intoxication evidence in the remains of a 900-year-old corpse]
- ^ One or more of the preceding sentences incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Dies, Christoph Albert". Encyclopædia Britannica. 8 (11th ed.). Cambridge University Press. p. 211.
- ^ Dies, Albert Christoph (1810). Biographische Nachrichten von Joseph Haydn nach mündlichen Erzählungen desselben entworfen und herausgegeben [Biographical Accounts of Joseph Haydn, written and edited from his own spoken narratives]. Vienna: Camesinaische Buchhandlung. English translation in: Dies, Albert Christoph (1963). "Biographical Accounts of Joseph Haydn". In Gotwals, Vernon (ed.). Haydn: Two Contemporary Portraits. (translation by Vernon Gotwals). Milwaukee: Univ. of Wisconsin Press. ISBN 0-299-02791-0.
- ^ "Beethoven und Blei: Tödliches Zusammenspiel". Archived from the original on 2009-02-21. Retrieved 2009-09-12.
- ^ "Beethoven litt unter Bleivergiftung". Archived from the original on 2009-02-21. Retrieved 2020-02-24.
- ^ Mary Seacole: Wonderful Adventures of Mrs. Seacole in Many Lands, Chapter IV, (1990 Oxford University Press reprint) ISBN 0-19-506672-3; (2005 Penguin 20th Century Classics reprint, ed. Sarah Salih) ISBN 0-14-043902-1
- ^ Jane Robinson: Mary Seacole: The Charismatic Black Nurse who became a heroine of the Crimea, p.53. Constable 2004 (p/b. ISBN 1-84119-677-0)
- ^ Weekly Irish Times, Saturday 15 October 1887; Enniskillen Chronicle and Erne Packet, 24 October 1887, p. 3
- ^ Gunn, Fenja. (1973). The Artificial Face: A History of Cosmetics. — as cited in Leisure Activities of an 18th Century Lady
- ^ Lead Based Hair Products: Too Hazardous for Household Use - Results, Howard W. Mielke, PhD, Myiesha D. Taylor, Chris R. Gonzales, M. Kelley Smith, Pamela V. Daniels, and Ayanna V.Buckner. Journal of American Pharmaceutical Association (NS37, Jan/Feb 1997:85-89).
- ^ Jump up to: a b "Can West News Service: Grecian Formula in a grey zone after ban". Archived from the original on 2007-08-08.
- ^ "The Proposition 65 List". Archived (PDF) from the original on 2014-10-31. Retrieved 2014-11-01.
- ^ The American Frugal Housewife, by Lydia M. Child
- ^ Laboratory manual in biology. Sharpe. 1911, American Book Company. p. 351
- ^ Andés, Louis Edgar, and Arthur Morris. Oil colours and printers' inks a practical handbook treating of linseed oil, boiled oil, paints, artists' colours, lampblack and printers' inks, black and coloured. London: Scott, Greenwood. 1903. 41. Print.
External links[]
| Wikimedia Commons has media related to Lead(II) acetate. |
- Case Studies in Environmental Medicine - Lead Toxicity
- Essay on "Lead Poisoning and Rome"
- HowStuffWorks "What Kind of Hair Color Do Men Use?" discussion of progressive dyes containing lead acetate
- National Pollutant Inventory - Lead and Lead Compounds Fact sheet (Does Not Bring Up Lead)
- ToxFAQs: Lead
- US Food and Drug Administration (FDA) fact sheet "Lead Acetate in Hair Dye Products"
- US Food and Drug Administration (FDA)21CFR73.2396 "PART 73 -- LISTING OF COLOR ADDITIVES EXEMPT FROM CERTIFICATION, Subpart C--Cosmetics, Sec. 73.2396 Lead acetate"
- Acetates
- Alchemical substances
- Lead(II) compounds
- Sugar substitutes