Ruthenium(III) acetate

| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C12H28BF4O18Ru3 | |
| Molar mass | 850.35 g·mol−1 |
| Appearance | green solid |
| Density | 2.110 g/cm3 |
| Structure | |
| octahedral | |
| Hazards | |
| GHS labelling: | |
 
| |
Signal word
|
Danger |
| H318, H410 | |
| P273, P280, P305+P351+P338, P310, P391, P501 | |
| Related compounds | |
Related compounds
|
Manganese(III) acetate Iron(III) acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ruthenium(III) acetate, commonly known as basic ruthenium acetate,[1] describes a family of salts where the cation has the formula [Ru3O(O2CCH3)6(OH2)3]+. A representative derivative is the dihydrate of the tetrafluoroborate salt [Ru3O(O2CCH3)6(OH2)3]BF4(H2O)2, which is the source of the data in the table above.[2] This and related salts are forest green, air-stable solids that are soluble in alcohols.
Basic ruthenium acetate features octahedral Ru(III) centers, a triply bridging oxo ligand, six acetate ligands, and three aquo ligands. The same structure is shared with basic acetates of iron, chromium, iridium, and manganese.[1][2]
Preparation and reactions[]
It is prepared by heating ruthenium trichloride in acetic acid in the presence of sodium acetate.[3] The basic acetates of ruthenium were reported in the early 1950s but were not properly formulated.[4]
Basic ruthenium acetate reacts with many ligands such as triphenylphosphine and pyridine concomitant with reduction. These derivatives [Ru3O(O2CCH3)6L3]0 are mixed valence compounds.[5]
Related compounds[]
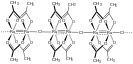 [Ru2(OAc)4Cl]n is a coordination polymer with a composition similar to that of ruthenium(III) acetate.
[Ru2(OAc)4Cl]n is a coordination polymer with a composition similar to that of ruthenium(III) acetate.
References[]
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ a b O. Almog, A. Bino, D. Garfinkel-Shweky (1993). "The Structure of Oxo-Bridged Trinuclear Ruthenium and Iridium Hexacarboxylates". Inorg. Chim. Acta. 213 (1–2): 99. doi:10.1016/S0020-1693(00)83819-0.CS1 maint: uses authors parameter (link)
- ^ J. C. Goeltz, S. D. Glover, J. Hauk, C. P. Kubiak (2010). Basic Ruthenium Acetate and Mixed Valence Derivatives. Inorg. Synth. Inorganic Syntheses. 35. pp. 156–160. doi:10.1002/9780470651568.ch8. ISBN 9780470651568.CS1 maint: uses authors parameter (link)
- ^ Martin, F. S. (1952). "Basic Trinuclear Ruthenium Acetate". Journal of the Chemical Society: 2682–4. doi:10.1039/jr9520002682.
- ^ Cotton, F. A.; Norman, J. G., Jr. (1972). "Structural Characterization of a Basic trinuclear Ruthenium Acetate". Inorg. Chim. Acta. 6: 411–419. doi:10.1016/S0020-1693(00)91829-2.CS1 maint: uses authors parameter (link)
- Ruthenium compounds
- Acetates
