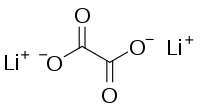
Lithium oxalate
| |
| Names | |
|---|---|
| Other names | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.008.232 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C 2Li 2O 4 | |
| Molar mass | 102.0 g/mol |
| Appearance | Colorless crystalline solid |
| Density | 2.12 g/cm3 |
| Hazards | |
| GHS labelling: | |

| |
Signal word
|
Warning |
| H302, H312 | |
| P264, P270, P280, P301+P312, P302+P352, P312, P322, P330, P363, P501 | |
| Related compounds | |
Related compounds
|
Calcium oxalate Sodium oxalate Magnesium oxalate Strontium oxalate Barium oxalate Potassium oxalate Beryllium oxalate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Lithium oxalate is an inorganic compound, a salt of lithium metal and oxalic acid with the chemical formula C
2Li
2O
4.[3][4] Lithium oxalate is highly insoluble in water and converts to the oxide when heated.[5]
Synthesis[]
One of the methods of synthesis is the reaction of direct neutralization of oxalic acid with lithium hydroxide:
Properties[]
The compound crystallizes in the monoclinic system, cell parameters a = 3.400, b = 5.156, c = 9.055 Å, β = 95.60°, Z = 4.[3]
Lithium oxalate decomposes when heated:
Applications[]
In pyrotechnics, the compound is used to color the flame red.[6]
References[]
- ^ "553-91-3 | Sigma-Aldrich". Sigma Aldrich. Retrieved 15 June 2021.
- ^ "di-Lithium oxalate". Merck Millipore. Retrieved 15 June 2021.
- ^ a b Beagley, B.; Small, R. W. H. (1964-06-10). "The structure of lithium oxalate". Acta Crystallographica. 17 (6): 783–788. doi:10.1107/S0365110X64002079. Retrieved 15 June 2021.
- ^ Solchenbach, Sophie; Wetjen, Morten; Pritzl, Daniel; Schwenke, K. Uta; Gasteiger, Hubert A. (2018). "Lithium Oxalate as Capacity and Cycle-Life Enhancer in LNMO/Graphite and LNMO/SiG Full Cells". Journal of the Electrochemical Society. 165 (3): A512–A524. doi:10.1149/2.0611803jes. Retrieved 15 June 2021.
- ^ "Lithium Oxalate". American Elements. Retrieved 15 June 2021.
- ^ Koch, Ernst-Christian (2009). Is it possible to Obtain a Deep Red Pyrotechnic Flame Based on Lithium?. 36th International Pyrotechnics Seminar. doi:10.13140/2.1.1657.0567. Retrieved 15 June 2021.
Categories:
- Inorganic compounds
- Lithium compounds
- Oxalates

