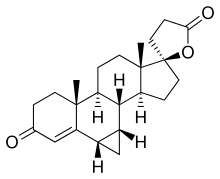
Prorenone Other names SC-23133; 3-(17β-Hydroxy-6β,7β-methylene-3-oxo-4-androsten-17α-yl)propionic acid γ-lactone ATC code
(1aS ,5aR ,5bS ,7aS ,8R ,10aS ,10bR ,10cS )-5a,7a-Dimethyl-1,1a,3',4,4',5,5a,5b,6,7,7a,9,10,10a,10b,10c-hexadecahydro-3H,5'H-spiro[cyclopenta[a]cyclopropa[l]phenanthrene-8,2'-furan]-3,5'-dione
CAS Number PubChem CID IUPHAR/BPS ChemSpider CompTox Dashboard (EPA ) Formula C 23 H 30 O 3 Molar mass −1 3D model (JSmol )
O=C6O[C@@]5([C@@]3([C@H]([C@@H]2[C@H]4[C@@H](/C1=C/C(=O)CC[C@]1(C)[C@H]2CC3)C4)CC5)C)CC6
InChI=1S/C23H30O3/c1-21-7-3-13(24)11-18(21)14-12-15(14)20-16(21)4-8-22(2)17(20)5-9-23(22)10-6-19(25)26-23/h11,14-17,20H,3-10,12H2,1-2H3/t14-,15+,16-,17-,20+,21+,22-,23+/m0/s1
Key:RRHHMFQGHCFGMH-LAPLKBAYSA-N
Prorenone (developmental code name SC-23133 ) is a steroidal antimineralocorticoid of the spirolactone group related to spironolactone that was never marketed.[1] lactonic form of prorenoic acid (prorenoate), and prorenoate potassium (SC-23992), the potassium salt of prorenoic acid, also exists.[1] potent than spironolactone as an antimineralocorticoid in animals, and it may act as a prodrug to prorenone.[1] mineralocorticoid receptor , prorenone also binds to the glucocorticoid , androgen , and progesterone receptors .[2] [3] in vivo [3] inhibitor of aldosterone biosynthesis .[4]
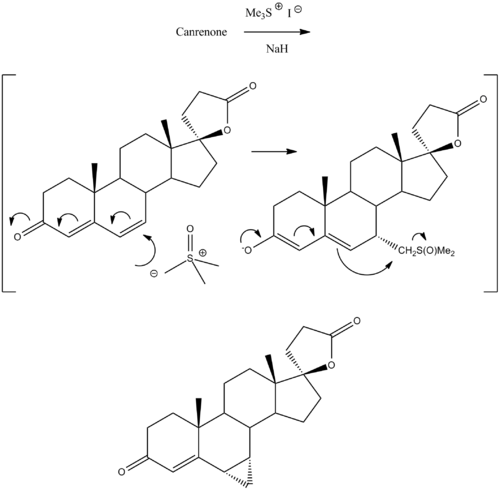
Chemistry [ ] Synthesis [ ] Prorenone can be synthesized via a Johnson–Corey–Chaykovsky reaction by reaction of canrenone with trimethylsulfonium iodide and sodium hydride .[5]
See also [ ] Canrenone Mexrenone Prorenoate potassium Prorenoic acid Potassium canrenoate References [ ]
^ a b c Claire, M.; Rafestin-Oblin, M. E.; Michaud, A.; Roth-Meyer, C.; Corvol, P. (1979). "Mechanism of Action of a New Antialdosterone Compound, Prorenone*". Endocrinology . 104 (4): 1194–1200. doi :10.1210/endo-104-4-1194 . ISSN 0013-7227 . PMID 436757 . ^ Gyorgy Szasz; Zsuzsanna Budvari-Barany (19 December 1990). Pharmaceutical Chemistry of Antihypertensive Agents ISBN 978-0-8493-4724-5 ^ a b Kamata S, Matsui T, Haga N, Nakamura M, Odaguchi K, Itoh T, Shimizu T, Suzuki T, Ishibashi M, Yamada F (September 1987). "Aldosterone antagonists. 2. Synthesis and biological activities of 11,12-dehydropregnane derivatives". J. Med. Chem . 30 (9): 1647–58. doi :10.1021/jm00392a022 . PMID 3040999 . ^ Netchitailo, Pierre; Delarue, Catherine; Perroteau, Isabelle; Leboulenger, Francois; Capron, Michel-Hubert; Vaudry, Hubert (1985). "Relative inhibitory potency of five mineralocorticoid antagonists on aldosterone biosynthesis in vitro". Biochemical Pharmacology . 34 (2): 189–194. doi :10.1016/0006-2952(85)90123-6 . ISSN 0006-2952 . PMID 2981534 . ^ Chinn, L.; 1974, U.S. Patent 3,845,041
AR
Agonists
Testosterone derivatives: 4-Androstenediol 4-Dehydroepiandrosterone (4-DHEA) 4-Hydroxytestosterone 5-Androstenedione 11-Ketotestosterone 11β-Hydroxyandrostenedione Adrenosterone (11-ketoandrostenedione, 11-oxoandrostenedione) Androstenediol (5-androstenediol)
Androstenedione (4-androstenedione) Atamestane Boldenone
Boldione (1,4-androstadienedione) Clostebol
Clostebol acetate Clostebol caproate Clostebol propionate Cloxotestosterone
Cloxotestosterone acetate Dehydroandrosterone DHEA (androstenolone, prasterone; 5-DHEA)
DHEA enanthate (prasterone enanthate) DHEA sulfate Exemestane Formestane Plomestane Quinbolone Silandrone Testosterone # (+dutasteride )
17α-Ethynyl-19-nortestosterone derivatives: Δ4 -Tibolone Desogestrel Etonogestrel Etynodiol
Gestodene Gestrinone Levonorgestrel Levonorgestrel esters (e.g., levonorgestrel butanoate )Lynestrenol
Lynestrenol phenylpropionate Norethisterone Norethisterone esters (e.g., norethisterone acetate , norethisterone enanthate )Norgestrel Norgestrienone Quingestanol
Tibolone MixedSARMs
Steroidal: MK-0773 TFM-4AS-1 YK-11 Antagonists
Nonsteroidal: 5N -Bicalutamide AA560 Apalutamide Atraric acid Bakuchiol Bicalutamide Bisphenols (e.g., BADGE , , bisphenol A , bisphenol F , bisphenol S )BMS-641988 Cimetidine Cioteronel Cyanonilutamide Darolutamide DDT (via metabolite p,p’-DDE )Dieldrin DIMP Endosulfan Enzalutamide EPI-001 EPI-7386 Fenarimol Flutamide Hydroxyflutamide Inocoterone Inocoterone acetate Ketoconazole Ketodarolutamide Lavender oil LG-120907 Linuron Methiocarb N -DesmethylapalutamideN -DesmethylenzalutamideNilutamide Pentomone Phenothrin Prochloraz Procymidone Proxalutamide Ralaniten (EPI-002) Ralaniten acetate (EPI-506) RD-162 Rezvilutamide RU-22930 RU-56187 RU-57073 RU-58642 RU-58841 Seviteronel Thalidomide Topilutamide (fluridil) Valproic acid Vinclozolin
GPRC6A
Agonists
Cations (incl. aluminum , calcium , gadolinium , magnesium , strontium , zinc )Dehydroandrosterone Dihydrotestosterone Estradiol L -α-Amino acidsL -arginineL -lysineL -ornithineOsteocalcin SHBG Testosterone
See also
Receptor/signaling modulators Androgens and antiandrogens Estrogen receptor modulators Progesterone receptor modulators List of androgens/anabolic steroids
MR
Agonists
11-Dehydrocorticosterone (11-oxocorticosterone, 17-deoxycortisone)
11-Deoxycorticosterone (desoxycortone, deoxycortone, desoxycorticosterone)
11-Deoxycortisol (cortodoxone, cortexolone)
Cortifen (cortiphen, kortifen) 11β-Hydroxyprogesterone 18-Hydroxy-11-deoxycorticosterone 19-Norprogesterone Aldosterone Corticosterone
Cortisol (hydrocortisone )
Cortisone
Fludrocortisone (fludrocortone)
Mometasone
Prednisolone
Prednisone
Antagonists
Nonsteroidal: Amlodipine Apararenone Benidipine Esaxerenone Felodipine Finerenone Nifedipine Nimodipine Nitrendipine
See also
Receptor/signaling modulators Mineralocorticoids and antimineralocorticoids Glucocorticoid receptor modulators List of corticosteroids
PR
Agonists
Testosterone derivatives: Progestins: 6,6-Difluoronorethisterone 6,6-Difluoronorethisterone acetate 17α-Allyl-19-nortestosterone Allylestrenol Altrenogest Chloroethynylnorgestrel Cingestol Danazol Desogestrel Dienogest Ethinylandrostenediol
Ethisterone Ethynerone Etonogestrel Etynodiol Etynodiol diacetate Gestodene Gestrinone Levonorgestrel Levonorgestrel esters (e.g., levonorgestrel butanoate )Lynestrenol Lynestrenol phenylpropionate Metynodiol Metynodiol diacetate Norelgestromin Norethisterone (norethindrone) Norethisterone esters (e.g., norethisterone acetate , norethisterone enanthate )Noretynodrel Norgesterone Norgestimate Norgestrel Norgestrienone Norvinisterone Oxendolone Quingestanol Quingestanol acetate Tibolone Tigestol Tosagestin ; Anabolic–androgenic steroids: 11β-Methyl-19-nortestosterone 11β-Methyl-19-nortestosterone dodecylcarbonate 19-Nor-5-androstenediol 19-Nor-5-androstenedione 19-Nordehydroepiandrosterone Bolandiol Bolandiol dipropionate Bolandione Dimethisterone Dienedione Dienolone Dimethandrolone Dimethandrolone buciclate Dimethandrolone dodecylcarbonate Dimethandrolone undecanoate Dimethyldienolone Dimethyltrienolone Ethyldienolone Ethylestrenol (ethylnandrol) Methyldienolone Metribolone (R-1881) Methoxydienone (methoxygonadiene) Mibolerone Nandrolone Nandrolone esters (e.g., nandrolone decanoate , nandrolone phenylpropionate )Norethandrolone Normethandrone (methylestrenolone, normethandrolone, normethisterone) RU-2309 Tetrahydrogestrinone Trenbolone (trienolone) Trenbolone esters (e.g., trenbolone acetate , trenbolone enanthate )Trendione Trestolone Trestolone acetate Spirolactone derivatives: Canrenoic acid Canrenone Drospirenone Mespirenone Potassium canrenoate Prorenone SC-5233 (spirolactone) SC-8109 Spironolactone Spirorenone MixedSPRMs
Nonsteroidal: Apigenin Kaempferol Naringenin Syringic acid Antagonists
Nonsteroidal: Darolutamide Valproic acid
mPR PAQR
See also
Receptor/signaling modulators Progestogens and antiprogestogens Androgen receptor modulators Estrogen receptor modulators List of progestogens