Boron trioxide
![Crystal structure of B2O3 [1]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/d9/B2O3powder.JPG/220px-B2O3powder.JPG)
| |
| |
| Names | |
|---|---|
| Other names
boron oxide, diboron trioxide, boron sesquioxide, boric oxide, boria
Boric acid anhydride | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.013.751 |
| EC Number |
|
| 11108 | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
Chemical formula
|
B2O3 |
| Molar mass | 69.6182 g/mol |
| Appearance | white, glassy solid |
| Density | 2.460 g/cm3, liquid; 2.55 g/cm3, trigonal; |
| Melting point | 450 °C (842 °F; 723 K) (trigonal) 510 °C (tetrahedral) |
| Boiling point | 1,860 °C (3,380 °F; 2,130 K) ,[2] sublimes at 1500 °C[3] |
| 1.1 g/100mL (10 °C) 3.3 g/100mL (20 °C) 15.7 g/100mL (100 °C) | |
| Solubility | partially soluble in methanol |
| Acidity (pKa) | ~ 4 |
Magnetic susceptibility (χ)
|
-39.0·10−6 cm3/mol |
| Thermochemistry | |
Heat capacity (C)
|
66.9 J/mol K |
Std molar
entropy (S |
80.8 J/mol K |
Std enthalpy of
formation (ΔfH⦵298) |
-1254 kJ/mol |
Gibbs free energy (ΔfG˚)
|
-832 kJ/mol |
| Hazards | |
| Main hazards | Irritant[4] |
| Safety data sheet | See: data page |
| GHS pictograms | 
|
| GHS Signal word | Danger |
GHS hazard statements
|
H360FD |
| P201, P202, P281, P308+313, P405, P501 | |
| NFPA 704 (fire diamond) | 
2
0
0 |
| Flash point | noncombustible |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
3163 mg/kg (oral, mouse)[5] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 15 mg/m3[4] |
REL (Recommended)
|
TWA 10 mg/m3[4] |
IDLH (Immediate danger)
|
2000 mg/m3[4] |
| Supplementary data page | |
Structure and
properties |
Refractive index (n), Dielectric constant (εr), etc. |
Thermodynamic
data |
Phase behaviour solid–liquid–gas |
Spectral data
|
UV, IR, NMR, MS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Boron trioxide (or diboron trioxide) is one of the oxides of boron. It is a white, glassy solid with the formula B2O3. It is almost always found as the vitreous (amorphous) form; however, it can be crystallized after extensive annealing (that is, under prolonged heat).
Glassy boron oxide (g-B2O3) is thought to be composed of which are six-membered rings composed of alternating 3-coordinate boron and 2-coordinate oxygen. Because of the difficulty of building disordered models at the correct density with many , this view was initially controversial, but such models have recently been constructed and exhibit properties in excellent agreement with experiment.[6] It is now recognized, from experimental and theoretical studies,[7][8][9][10][11] that the fraction of boron atoms belonging to boroxol rings in glassy B2O3 is somewhere between 0.73 and 0.83, with 0.75 (3⁄4) corresponding to a 1:1 ratio between ring and non-ring units. The number of boroxol rings decays in the liquid state with increasing temperature.[12]
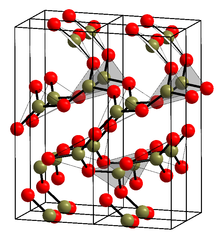
The crystalline form (α-B2O3) (see structure in the infobox[1]) is exclusively composed of BO3 triangles. This trigonal, quartz-like network undergoes a coesite-like transformation to monoclinic β-B2O3 at several gigapascals (9.5 GPa).[13]
Preparation[]
Boron trioxide is produced by treating borax with sulfuric acid in a . At temperatures above 750 °C, the molten boron oxide layer separates out from sodium sulfate. It is then decanted, cooled and obtained in 96–97% purity.[3]
Another method is heating boric acid above ~300 °C. Boric acid will initially decompose into steam, (H2O(g)) and metaboric acid (HBO2) at around 170 °C, and further heating above 300 °C will produce more steam and diboron trioxide. The reactions are:
- H3BO3 → HBO2 + H2O
- 2 HBO2 → B2O3 + H2O
Boric acid goes to anhydrous microcrystalline B2O3 in a heated fluidized bed.[14] Carefully controlled heating rate avoids gumming as water evolves. Molten boron oxide attacks silicates. Internally graphitized tubes via acetylene thermal decomposition are passivated.[15]
Crystallization of molten α-B2O3 at ambient pressure is strongly kinetically disfavored (compare liquid and crystal densities). Threshold conditions for crystallization of the amorphous solid are 10 kbar and ~200 °C.[16] Its proposed crystal structure in enantiomorphic space groups P31(#144); P32(#145)[17][18] (e.g., γ-glycine) has been revised to enantiomorphic space groups P3121(#152); P3221(#154)[19](e.g., α-quartz).
Boron oxide will also form when diborane (B2H6) reacts with oxygen in the air or trace amounts of moisture:
- 2B2H6(g) + 3O2(g) → 2B2O3(s) + 6H2(g)
- B2H6(g) + 3H2O(g) → B2O3(s) + 6H2(g)[20]
Applications[]
- Fluxing agent for glass and enamels
- Starting material for synthesizing other boron compounds such as boron carbide
- An additive used in glass fibres (optical fibres)
- Component used in the production of borosilicate glass
- The inert capping layer in the Liquid Encapsulation Czochralski process for the production of gallium arsenide single crystal
- As an acid catalyst in organic synthesis
See also[]
References[]
- ^ Jump up to: a b Gurr, G. E.; Montgomery, P. W.; Knutson, C. D.; Gorres, B. T. (1970). "The Crystal Structure of Trigonal Diboron Trioxide". Acta Crystallographica B. 26 (7): 906–915. doi:10.1107/S0567740870003369.
- ^ High temperature corrosion and materials chemistry: proceedings of the Per Kofstad Memorial Symposium. Proceedings of the Electrochemical Society. The Electrochemical Society. 2000. p. 496. ISBN 978-1-56677-261-7.
- ^ Jump up to: a b Patnaik, P. (2003). Handbook of Inorganic Chemical Compounds. McGraw-Hill. p. 119. ISBN 978-0-07-049439-8. Retrieved 2009-06-06.
- ^ Jump up to: a b c d NIOSH Pocket Guide to Chemical Hazards. "#0060". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Boron oxide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Ferlat, G.; Charpentier, T.; Seitsonen, A. P.; Takada, A.; Lazzeri, M.; Cormier, L.; Calas, G.; Mauri. F. (2008). "Boroxol Rings in Liquid and Vitreous B2O3 from First Principles". Phys. Rev. Lett. 101 (6): 065504. Bibcode:2008PhRvL.101f5504F. doi:10.1103/PhysRevLett.101.065504. PMID 18764473.; Ferlat, G.; Seitsonen, A. P.; Lazzeri, M.; Mauri, F. (2012). "Hidden polymorphs drive vitrification in B2O3". Nature Materials Letters. 11 (11): 925–929. arXiv:1209.3482. Bibcode:2012NatMa..11..925F. doi:10.1038/NMAT3416. PMID 22941329. S2CID 11567458.
- ^ Hung, I.; et al. (2009). "Determination of the bond-angle distribution in vitreous B2O3 by rotation (DOR) NMR spectroscopy". Journal of Solid State Chemistry. 182 (9): 2402–2408. Bibcode:2009JSSCh.182.2402H. doi:10.1016/j.jssc.2009.06.025.
- ^ Soper, A. K. (2011). "Boroxol rings from diffraction data on vitreous boron trioxide". J. Phys.: Condens. Matter. 23 (36): 365402. Bibcode:2011JPCM...23.5402S. doi:10.1088/0953-8984/23/36/365402. PMID 21865633.
- ^ Joo, C.; et al. (2000). "The ring structure of boron trioxide glass". Journal of Non-Crystalline Solids. 261 (1–3): 282–286. Bibcode:2000JNCS..261..282J. doi:10.1016/s0022-3093(99)00609-2.
- ^ Zwanziger, J. W. (2005). "The NMR response of boroxol rings: a density functional theory study". Solid State Nuclear Magnetic Resonance. 27 (1–2): 5–9. doi:10.1016/j.ssnmr.2004.08.004. PMID 15589722.
- ^ Micoulaut, M. (1997). "The structure of vitreous B2O3 obtained from a thermostatistical model of agglomeration". Journal of Molecular Liquids. 71 (2–3): 107–114. doi:10.1016/s0167-7322(97)00003-2.
- ^ Alderman, O. L. G. Ferlat, G. Baroni, A. Salanne, M. Micoulaut, M. Benmore, C. J. Lin, A. Tamalonis, A. Weber, J. K. R. (2015). "Liquid B2O3 up to 1700K: X-ray diffraction and boroxol ring dissolution" (PDF). Journal of Physics: Condensed Matter. 27 (45): 455104. doi:10.1088/0953-8984/27/45/455104. PMID 26499978.CS1 maint: multiple names: authors list (link)
- ^ Brazhkin, V. V.; Katayama, Y.; Inamura, Y.; Kondrin, M. V.; Lyapin, A. G.; Popova, S. V.; Voloshin, R. N. (2003). "Structural transformations in liquid, crystalline and glassy B2O3 under high pressure". JETP Letters. 78 (6): 393–397. Bibcode:2003JETPL..78..393B. doi:10.1134/1.1630134. S2CID 189764568.
- ^ Kocakuşak, S.; Akçay, K.; Ayok, T.; Koöroğlu, H. J.; Koral, M.; Savaşçi, Ö. T.; Tolun, R. (1996). "Production of anhydrous, crystalline boron oxide in fluidized bed reactor". Chemical Engineering and Processing. 35 (4): 311–317. doi:10.1016/0255-2701(95)04142-7.
- ^ Morelock, C. R. (1961). "Research Laboratory Report #61-RL-2672M". General Electric. Cite journal requires
|journal=(help) - ^ Aziz, M. J.; Nygren, E.; Hays, J. F.; Turnbull, D. (1985). "Crystal Growth Kinetics of Boron Oxide Under Pressure". Journal of Applied Physics. 57 (6): 2233. Bibcode:1985JAP....57.2233A. doi:10.1063/1.334368.
- ^ Gurr, G. E.; Montgomery, P. W.; Knutson, C. D.; Gorres, B. T. (1970). "The crystal structure of trigonal diboron trioxide". Acta Crystallographica B. 26 (7): 906–915. doi:10.1107/S0567740870003369.
- ^ Strong, S. L.; Wells, A. F.; Kaplow, R. (1971). "On the crystal structure of B2O3". Acta Crystallographica B. 27 (8): 1662–1663. doi:10.1107/S0567740871004515.
- ^ Effenberger, H.; Lengauer, C. L.; Parthé, E. (2001). "Trigonal B2O3 with Higher Space-Group Symmetry: Results of a Reevaluation". Monatshefte für Chemie. 132 (12): 1515–1517. doi:10.1007/s007060170008. S2CID 97795834.
- ^ AirProducts (2011). "Diborane Storage & Delivery" (PDF). Archived from the original (PDF) on 2015-02-04. Retrieved 2013-08-21. Cite journal requires
|journal=(help)
External links[]
- Boron compounds
- Acidic oxides
- Glass compositions
- Sesquioxides