Indium(III) iodide
| Names | |
|---|---|
| Other names
Indium triiodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.475 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| InI3 | |
| Molar mass | 495.53 |
| Appearance | Yellow solid |
| Density | 4.69 g/cm3 |
| Melting point | 210 °C (410 °F; 483 K) |
| Boiling point | 500 °C (932 °F; 773 K) |
| Related compounds | |
Other anions
|
Indium(III) bromide Indium(III) chloride |
Other cations
|
Aluminum iodide Gallium(III) iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Indium(III) iodide or indium triiodide is a chemical compound of indium and iodine with the formula InI3.
Preparation[]
Indium(III) iodide is obtained by evaporation of a solution of indium in HI.[1]
Properties[]
Distinct yellow and a red forms are known. The red form undergoes a transition to the yellow at 57 °C. The structure of the red form has not been determined by X-ray crystallography, however spectroscopic evidence indicates that indium may be six coordinate.[2] The yellow form consists of In2I6 with 4 coordinate indium centres.
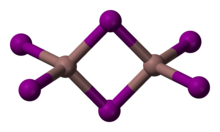
Ball-and-stick model of the In2I6 molecule
References[]
Categories:
- Iodides
- Indium compounds
- Metal halides
- Chemistry stubs