Ascorbate peroxidase
| L-ascorbate peroxidase | |||
|---|---|---|---|
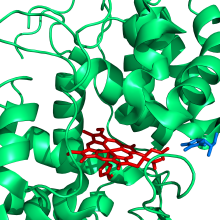 Structure of ascorbate peroxidase in complex with ascorbate (in blue); a histidine ligand (in red) coordinates to the iron of the heme group (also in red). Image taken from PDB 1OAF and created using Pymol | |||
| Identifiers | |||
| EC no. | 1.11.1.11 | ||
| CAS no. | 72906-87-7 | ||
| Databases | |||
| IntEnz | IntEnz view | ||
| BRENDA | BRENDA entry | ||
| ExPASy | NiceZyme view | ||
| KEGG | KEGG entry | ||
| MetaCyc | metabolic pathway | ||
| PRIAM | profile | ||
| PDB structures | RCSB PDB PDBe PDBsum | ||
| Gene Ontology | AmiGO / QuickGO | ||
| |||
Ascorbate peroxidase (or L-ascorbate peroxidase, APX) (EC 1.11.1.11) is an enzyme that catalyzes the chemical reaction
- L-ascorbate + H2O2 dehydroascorbate + 2 H2O
It is a member of the family of heme-containing peroxidases. Heme peroxidases catalyse the H2O2-dependent oxidation of a wide range of different, usually organic, substrates in biology.
This enzyme belongs to the family of oxidoreductases, specifically those acting on a peroxide as acceptor (peroxidases). The systematic name of this enzyme class is L-ascorbate:hydrogen-peroxide oxidoreductase. Other names in common use include L-ascorbic acid peroxidase, L-ascorbic acid-specific peroxidase, ascorbate peroxidase, and ascorbic acid peroxidase. This enzyme participates in ascorbate and aldarate metabolism.
Overview[]
Ascorbate-dependent peroxidase activity was first reported in 1979,[1],[2] more than 150 years after the first observation of peroxidase activity in horseradish plants[3] and almost 40 years after the discovery of the closely related cytochrome c peroxidase enzyme.[4]
Peroxidases have been classified into three types (class I, class II and class III): ascorbate peroxidases is a class I peroxidase enzyme.[5] APXs catalyse the H2O2-dependent oxidation of ascorbate in plants, algae and certain cyanobacteria.[6] APX has high sequence identity to cytochrome c peroxidase, which is also a class I peroxidase enzyme. Under physiological conditions, the immediate product of the reaction, the monodehydroascorbate radical, is reduced back to ascorbate by a monodehydroascorbate reductase (monodehydroascorbate reductase (NADH)) enzyme. In the absence of a reductase, two monodehydroascorbate radicals disproportionate rapidly to dehydroascorbic acid and ascorbate. APX is an integral component of the glutathione-ascorbate cycle.[7]
Substrate specificity[]
APX enzymes show high specificity for ascorbate as an electron donor, but most APXs will also oxidise other organic substrates that are more characteristic of the class III peroxidases (such as horseradish peroxidase), in some cases at rates comparable to that of ascorbate itself. This means that defining an enzyme as an APX is not straightforward, but is usually applied when the specific activity for ascorbate is higher than that for other substrates.
Mechanism[]
Most of the information on mechanism comes from work on the pea cytosolic and soybean cytosolic enzymes. The mechanism of oxidation of ascorbate is achieved by means of an oxidized Compound I intermediate, which is subsequently reduced by substrate in two, sequential single electron transfer steps (equations [1]–[3], where HS = substrate and S• = one electron oxidised form of substrate).
- APX + H2O2 → Compound I + H2O [1]
- Compound I + HS → Compound II + S• [2]
- Compound II + HS → APX + S• + H2O [3]
In ascorbate peroxidase, Compound I is a transient (green) species and contains a high-valent iron species (known as ferryl heme, FeIV) and a porphyrin pi-cation radical,[8],[9] as found in horseradish peroxidase. Compound II contains only the ferryl heme.
Structural information[]
The structure of pea cytosolic APX was reported in 1995.[10] The binding interaction of soybean cytosolic APX with its physiological substrate, ascorbate[11],[12] and with a number of other substrates[13] are also known.
As of late 2007, 12 structures have been solved for this class of enzymes, with PDB accession codes 1APX, 1IYN, 1OAF, 1OAG, 1V0H, 2CL4, 2GGN, 2GHC, 2GHD, 2GHE, 2GHH, and 2GHK.
Applications in cellular imaging[]
Both pea APX[14] and soybean APX[15] have been used in electron microscopy studies for cellular imaging.
See also[]
References[]
- ^ Kelly GJ, Latzko E (December 1979). "Soluble ascorbate peroxidase: detection in plants and use in vitamim C estimation". Die Naturwissenschaften. 66 (12): 617–9. doi:10.1007/bf00405128. PMID 537642. S2CID 12729653.
- ^ Groden D, Beck E (June 1979). "H2O2 destruction by ascorbate-dependent systems from chloroplasts". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 546 (3): 426–35. doi:10.1016/0005-2728(79)90078-1. PMID 454577.
- ^ Planche LA (1810). "Note sur la sophistication de la résine de jalap et sur les moyens de la reconnaître". Bull Pharm. 2: 578–80.
- ^ Altschul AM, Abrams R, Hogness TR (1940). "Cytochrome c Peroxidase" (PDF). Journal of Biological Chemistry. 136 (3): 777–794. doi:10.1016/S0021-9258(18)73036-6.
- ^ Welinder KG (1992). "Superfamily of plant, fungal and bacterial peroxidases". Curr. Opin. Chem. Biol. 2 (3): 388–393. doi:10.1016/0959-440x(92)90230-5.
- ^ Raven EL (August 2003). "Understanding functional diversity and substrate specificity in haem peroxidases: what can we learn from ascorbate peroxidase?". Natural Product Reports. 20 (4): 367–81. doi:10.1039/B210426C. PMID 12964833.
- ^ Noctor G, Foyer CH (June 1998). "ASCORBATE AND GLUTATHIONE: Keeping Active Oxygen Under Control". Annual Review of Plant Physiology and Plant Molecular Biology. 49: 249–279. doi:10.1146/annurev.arplant.49.1.249. PMID 15012235.
- ^ Patterson WR, Poulos TL, Goodin DB (April 1995). "Identification of a porphyrin pi cation radical in ascorbate peroxidase compound I". Biochemistry. 34 (13): 4342–5. doi:10.1021/bi00013a024. PMID 7703248.
- ^ Jones DK, Dalton DA, Rosell FI, Raven EL (December 1998). "Class I heme peroxidases: characterization of soybean ascorbate peroxidase". Archives of Biochemistry and Biophysics. 360 (2): 173–8. doi:10.1006/abbi.1998.0941. PMID 9851828.
- ^ Patterson WR, Poulos TL (April 1995). "Crystal structure of recombinant pea cytosolic ascorbate peroxidase". Biochemistry. 34 (13): 4331–41. doi:10.1021/bi00013a023. PMID 7703247.
- ^ Sharp KH, Mewies M, Moody PC, Raven EL (April 2003). "Crystal structure of the ascorbate peroxidase-ascorbate complex". Nature Structural Biology. 10 (4): 303–7. doi:10.1038/nsb913. PMID 12640445. S2CID 32035409.
- ^ Macdonald IK, Badyal SK, Ghamsari L, Moody PC, Raven EL (June 2006). "Interaction of ascorbate peroxidase with substrates: a mechanistic and structural analysis". Biochemistry. 45 (25): 7808–17. doi:10.1021/bi0606849. PMID 16784232.
- ^ Gumiero A, Murphy EJ, Metcalfe CL, Moody PC, Raven EL (August 2010). "An analysis of substrate binding interactions in the heme peroxidase enzymes: a structural perspective". Archives of Biochemistry and Biophysics. 500 (1): 13–20. doi:10.1016/j.abb.2010.02.015. PMID 20206594.
- ^ Martell JD, Deerinck TJ, Sancak Y, Poulos TL, Mootha VK, Sosinsky GE, et al. (November 2012). "Engineered ascorbate peroxidase as a genetically encoded reporter for electron microscopy". Nature Biotechnology. 30 (11): 1143–8. doi:10.1038/nbt.2375. PMC 3699407. PMID 23086203.
- ^ Lam SS, Martell JD, Kamer KJ, Deerinck TJ, Ellisman MH, Mootha VK, Ting AY (January 2015). "Directed evolution of APEX2 for electron microscopy and proximity labeling". Nature Methods. 12 (1): 51–4. doi:10.1038/nmeth.3179. PMC 4296904. PMID 25419960.
Further reading[]
- Shigeoka S, Nakano Y, Kitaoka S (April 1980). "Purification and some properties of L-ascorbic-acid-specific peroxidase in Euglena gracilis Z". Archives of Biochemistry and Biophysics. 201 (1): 121–7. doi:10.1016/0003-9861(80)90495-6. PMID 6772104.
- Shigeoka S, Nakano Y, Kitaoka S (January 1980). "Metabolism of hydrogen peroxide in Euglena gracilis Z by L-ascorbic acid peroxidase". The Biochemical Journal. 186 (1): 377–80. doi:10.1042/bj1860377. PMC 1161541. PMID 6768357.
External links[]
- EC 1.11.1.11 Archived 2011-05-16 at the Wayback Machine
- L-ascorbate peroxidase (EC-Number 1.11.1.11 )
- EC 1.11.1
- Enzymes of known structure
