GMP synthase
| GMP synthetase (glutamine-hydrolyzing) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 GMP synthetase, human | |||||||||
| Identifiers | |||||||||
| EC no. | 6.3.5.2 | ||||||||
| CAS no. | 37318-71-1 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| GMP synthetase C terminal domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
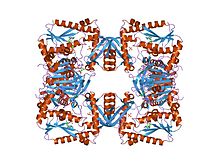 escherichia coli gmp synthetase complexed with amp and pyrophosphate.[1] | |||||||||
| Identifiers | |||||||||
| Symbol | GMP_synt_C | ||||||||
| Pfam | PF00958 | ||||||||
| InterPro | IPR001674 | ||||||||
| PROSITE | PDOC00405 | ||||||||
| SCOP2 | 1gpm / SCOPe / SUPFAM | ||||||||
| |||||||||
| GMPS | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||
| Aliases | GMPS, GMP synthase, guanine monophosphate synthase, GATD7, GMP synthase | ||||||||||||||||||||||||
| External IDs | OMIM: 600358 MGI: 2448526 HomoloGene: 68367 GeneCards: GMPS | ||||||||||||||||||||||||
| |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Orthologs | |||||||||||||||||||||||||
| Species | Human | Mouse | |||||||||||||||||||||||
| Entrez | |||||||||||||||||||||||||
| Ensembl | |||||||||||||||||||||||||
| UniProt | |||||||||||||||||||||||||
| RefSeq (mRNA) | |||||||||||||||||||||||||
| RefSeq (protein) | |||||||||||||||||||||||||
| Location (UCSC) | Chr 3: 155.87 – 155.94 Mb | Chr 3: 63.88 – 63.93 Mb | |||||||||||||||||||||||
| PubMed search | [4] | [5] | |||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
Guanosine monophosphate synthetase, (EC 6.3.5.2) also known as GMPS is an enzyme that converts xanthosine monophosphate to guanosine monophosphate.[6]
In the de novo synthesis of purine nucleotides, IMP is the branch point metabolite at which point the pathway diverges to the synthesis of either guanine or adenine nucleotides. In the guanine nucleotide pathway, there are 2 enzymes involved in converting IMP to GMP, namely IMP dehydrogenase (IMPD1), which catalyzes the oxidation of IMP to XMP, and GMP synthetase, which catalyzes the amination of XMP to GMP.[6]
Enzymology[]
In enzymology, a GMP synthetase (glutamine-hydrolysing) (EC 6.3.5.2) is an enzyme that catalyzes the chemical reaction
- ATP + xanthosine 5'-phosphate + L-glutamine + H2O AMP + diphosphate + GMP + L-glutamate
The 4 substrates of this enzyme are ATP, xanthosine 5'-phosphate, L-glutamine, and H2O, whereas its 4 products are AMP, diphosphate, GMP, and L-glutamate.
This enzyme belongs to the family of ligases, specifically those forming carbon-nitrogen bonds carbon-nitrogen ligases with glutamine as amido-N-donor. The systematic name of this enzyme class is xanthosine-5'-phosphate:L-glutamine amido-ligase (AMP-forming). Other names in common use include GMP synthetase (glutamine-hydrolysing), guanylate synthetase (glutamine-hydrolyzing), guanosine monophosphate synthetase (glutamine-hydrolyzing), xanthosine 5'-phosphate amidotransferase, and guanosine 5'-monophosphate synthetase. This enzyme participates in purine metabolism and glutamate metabolism. At least one compound, is known to inhibit this enzyme.
Structural studies[]
As of late 2007, 5 structures have been solved for this class of enzymes, with PDB accession codes 1GPM, 1WL8, 2A9V, 2D7J, and 2DPL.
Role in Metabolism[]
Purine Metabolism[]
GMP Synthase is the second step in the generation of GMP from IMP; the first step occurs when IMP dehydrogenase generates XMP, and then GMP synthetase is able to react with glutamine and ATP to generate GMP. IMP may also be generated into AMP by Adenylosuccinate synthetase and then adenylosuccinate lyase.[7]
Amino Acid Metabolism[]
GMP synthase is also involved in amino acid metabolism because it generates L-glutamate from L-glutamine.[7]
Organismal involvement[]
This enzyme is widely distributed and a number of crystal structures have been solved, including in Escherichia coli, Pyrococcus Horikoshii, Thermoplasma acidophil, Homo sapiens, Thermus thermophilus and Mycobacterium tuberculosis. The most extensive structural studies have been done in E. coli.[8]
Structure and Function[]
GMP synthase forms a tetramer in an open box shape, which is a dimer of dimers. The R interfaces are held together with a hydrophobic core and a beta sheet, while the P dimer interfaces do not have a hydrophobic core and are more variable than the R interfaces.[8] This enzyme also binds several ligands, including phosphate, pyrophosphate, AMP, citrate and Magnesium.[9]
Class I Amidotransferase Domain[]
The amidotransferase domain is responsible for removal of the amide nitrogen from the glutamine substrate. The class I amidotransferase domain is made of the N terminal 206 residues of the enzyme, and consists of 12 beta strands and 5 alpha helices; the core of this domain is an open 7-stranded mixed beta sheet. Its catalytic triad includes Cys86, His181 and Glu183. His181 is a base and Glu183 is a Hydrogen bond acceptor from the Histidine imidazole ring. Cys86 is the catalytic residue and is conserved. It falls into a nucleophile elbow, where it is at the end of a beta strand and the beginning of an alpha helix, and has little flexibility in its phi and psi angles; thus, Gly84 and Gly88 are conserved and allow for the tight packing of amino acids surrounding the catalytic residue.[8]
Synthetase Domain: ATP Pyrophosphatase domain[]
The synthetase domain is responsible for the addition of the abstracted Nitrogen to the acceptor substrate. The ATP Pyrophosphatase domain consists of a beta sheet containing 5 parallel strands with several alpha helices on each side. The P loop is the nucleotide binding motif; residues 235-241 make up the P loop which specifically binds to pyrophosphate.[8]
The structure of this domain is what creates the specificity of this enzyme for ATP. The binding pocket forms hydrophobic interactions with the adenine ring, and the backbone of Val260 forms H bonds with multiple Nitrogens in the ring of AMP, which excludes substituents on the C2 purine ring. This creates extreme specificity for adenine and ATP binding.[8]
References[]
- ^ Tesmer JJ, Klem TJ, Deras ML, Davisson VJ, Smith JL (January 1996). "The crystal structure of GMP synthetase reveals a novel catalytic triad and is a structural paradigm for two enzyme families". Nature Structural Biology. 3 (1): 74–86. doi:10.1038/nsb0196-74. PMID 8548458. S2CID 30864133.
- ^ a b c GRCh38: Ensembl release 89: ENSG00000163655 - Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000027823 - Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b "Entrez Gene: GMPS guanine monphosphate synthetase".
- ^ a b Garrett RH (1998). Biochemistry. [Place of publication not identified]: Harcourt College. ISBN 0-03-044857-3. OCLC 947935503.
- ^ a b c d e Tesmer JJ, Klem TJ, Deras ML, Davisson VJ, Smith JL (January 1996). "The crystal structure of GMP synthetase reveals a novel catalytic triad and is a structural paradigm for two enzyme families". Nature Structural Biology. 3 (1): 74–86. doi:10.1038/nsb0196-74. PMID 8548458.
- ^ "Ligand/metal interactions: 1gpm". www.ebi.ac.uk. Retrieved 2021-10-21.
Further reading[]
- Page T, Bakay B, Nyhan WL (1984). "Human GMP synthetase". The International Journal of Biochemistry. 16 (1): 117–20. doi:10.1016/0020-711X(84)90061-2. PMID 6698284.
- Nakamura J, Straub K, Wu J, Lou L (October 1995). "The glutamine hydrolysis function of human GMP synthetase. Identification of an essential active site cysteine". The Journal of Biological Chemistry. 270 (40): 23450–5. doi:10.1074/jbc.270.40.23450. PMID 7559506.
- Nakamura J, Lou L (March 1995). "Biochemical characterization of human GMP synthetase". The Journal of Biological Chemistry. 270 (13): 7347–53. doi:10.1074/jbc.270.13.7347. PMID 7706277.
- Hirst M, Haliday E, Nakamura J, Lou L (September 1994). "Human GMP synthetase. Protein purification, cloning, and functional expression of cDNA". The Journal of Biological Chemistry. 269 (38): 23830–7. PMID 8089153.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Fedorova L, Kost-Alimova M, Gizatullin RZ, Alimov A, Zabarovska VI, Szeles A, et al. (1997). "Assignment and ordering of twenty-three unique NotI-linking clones containing expressed genes including the guanosine 5'-monophosphate synthetase gene to human chromosome 3". European Journal of Human Genetics. 5 (2): 110–6. doi:10.1159/000484744. PMID 9195163.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Pegram LD, Megonigal MD, Lange BJ, Nowell PC, Rowley JD, Rappaport EF, Felix CA (December 2000). "t(3;11) translocation in treatment-related acute myeloid leukemia fuses MLL with the GMPS (GUANOSINE 5' MONOPHOSPHATE SYNTHETASE) gene". Blood. 96 (13): 4360–2. doi:10.1182/blood.V96.13.4360. PMID 11110714.
- Guo D, Han J, Adam BL, Colburn NH, Wang MH, Dong Z, et al. (December 2005). "Proteomic analysis of SUMO4 substrates in HEK293 cells under serum starvation-induced stress". Biochemical and Biophysical Research Communications. 337 (4): 1308–18. doi:10.1016/j.bbrc.2005.09.191. PMID 16236267.
- Abrams R, Bentley M (1959). "Biosynthesis of nucleic acid purines. III. Guanosine 5'-phosphate formation from xanthosine 5'-phosphate and L-glutamine". Arch. Biochem. Biophys. 79: 91–110. doi:10.1016/0003-9861(59)90383-2.
- Lagerkvist U (July 1958). "Biosynthesis of guanosine 5'-phosphate. II. Amination of xanthosine 5'-phosphate by purified enzyme from pigeon liver". The Journal of Biological Chemistry. 233 (1): 143–9. PMID 13563458.
External links[]
- GMP+synthetase at the US National Library of Medicine Medical Subject Headings (MeSH)
- PDBe-KB provides an overview of all the structure information available in the PDB for Human GMP synthase [glutamine-hydrolyzing]
- Genes on human chromosome 3
- EC 6.3.5
- Enzymes of known structure





