Gold(III) chloride
| |
 Ball-and-stick model of AuCl3
| |
 Crystal structure of AuCl3
| |
| Names | |
|---|---|
| IUPAC name
Gold(III) trichloride
| |
| Other names
Auric chloride
Gold trichloride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.033.280 |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| AuCl3 (exists as Au2Cl6) | |
| Molar mass | 606.6511 g/mol |
| Appearance | Red crystals (anhydrous); golden, yellow crystals (monohydrate)[1] |
| Density | 4.7 g/cm3 |
| Melting point | 254 °C (489 °F; 527 K) (decomposes) |
| 68 g/100 ml (cold) | |
| Solubility | soluble in ether and ethanol, slightly soluble in liquid ammonia |
Magnetic susceptibility (χ)
|
−112·10−6 cm3/mol |
| Structure | |
| monoclinic | |
| Square planar | |
| Hazards[2] | |
| Main hazards | Irritant |
| Safety data sheet | See: data page |
| GHS pictograms | 
|
| GHS Signal word | Warning |
GHS hazard statements
|
H315, H319, H335 |
| P261, P305+351+338 | |
| Related compounds | |
Other anions
|
Gold(III) fluoride Gold(III) bromide Gold(III) nitrate |
Other cations
|
Gold(I) chloride Silver(I) chloride Platinum(II) chloride Mercury(II) chloride |
| Supplementary data page | |
Structure and
properties |
Refractive index (n), Dielectric constant (εr), etc. |
Thermodynamic
data |
Phase behaviour solid–liquid–gas |
Spectral data
|
UV, IR, NMR, MS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Gold(III) chloride, traditionally called auric chloride, is a chemical compound of gold and chlorine with the molecular formula Au2Cl6. The "III" in the name indicates the gold has an oxidation state of +3, which is typical for gold compounds. Gold(III) chloride is hygroscopic and decomposes in visible light. This compound is a dimer, which means that two identical subunits comprise each molecule. This compound has a variety of uses, such as catalyzing a variety of organic reactions.
Structure[]
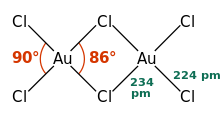
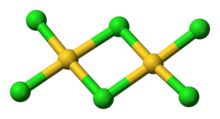
AuCl3 exists as a chloride-bridged dimer both as a solid and a vapour, at least at low temperatures.[3] Gold(III) bromide behaves analogously.[1] The structure is similar to that of iodine(III) chloride.
In gold(III) chloride, each gold centre is square planar,[1] which is typical of a metal complex with a d8 electron count. The bonding in AuCl3 is considered somewhat covalent.
Preparation[]
Gold(III) chloride is most often prepared by passing chlorine gas over gold powder at 180 °C (356 °F):[1]
- 2 Au + 3 Cl2 → Au2Cl6
The chlorination reaction can be conducted in the presence of tetrabutylammonium chloride, the product being the lipophilic salt tetrabutylammonium tetrachloraurate.[4]
Another method of preparation is via chloroauric acid, which is obtained by first dissolving the gold powder in aqua regia to give chloroauric acid:[5]
- Au + HNO3 + 4 HCl → HAuCl4 + 2 H2O + NO
The resulting chloroauric acid is subsequently heated to give Au2Cl6:
- 2 HAuCl4 → Au2Cl6 + 2 HCl
Reactions[]

On contact with water, AuCl
3 forms acidic hydrates and the conjugate base [AuCl
3(OH)]−
. An Fe2+
ion may reduce it, causing elemental gold to be precipitated from the solution.[1]
Anhydrous AuCl3 begins to decompose to AuCl at around 160 °C (320 °F), however, this, in turn, undergoes disproportionation at higher temperatures to give gold metal and AuCl3:
- AuCl3 → AuCl + Cl2 (>160 °C)
- 3 AuCl → AuCl3 + 2 Au (>420 °C)
AuCl3 is a lewis acid and readily forms complexes. For example, it reacts with hydrochloric acid to form chloroauric acid (HAuCl
4):
- HCl + AuCl
3 → H+
+ [AuCl
4]−
Chloroauric acid is the product formed when gold dissolves in aqua regia.
Other chloride sources, such as KCl, also convert AuCl3 into AuCl−
4. Aqueous solutions of AuCl3 react with an aqueous base such as sodium hydroxide to form a precipitate of Au(OH)3, which will dissolve in excess NaOH to form sodium aurate (NaAuO2). If gently heated, Au(OH)3 decomposes to gold(III) oxide, Au2O3, and then to gold metal.[6][7][8][9][10]
Gold(III) chloride is the starting point for the chemical synthesis of many other gold compounds. For example, the reaction with potassium cyanide produces the water-soluble complex, K[Au(CN)4]:
- AuCl
3 + 4 KCN → K[Au(CN)
4] + 3 KCl
Applications in organic synthesis[]
As of 2003, AuCl3 has attracted the interest of organic chemists as a mild acid catalyst for a variety of reactions, AuCl3,[11] although no transformations have been commercialised. Gold(III) salts, especially Na[AuCl4], provide an alternative to mercury(II) salts as catalysts for reactions involving alkynes. An illustrative reaction is the hydration of terminal alkynes to produce acetyl compounds.[12]
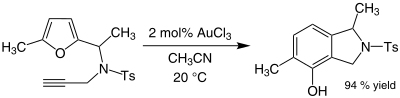
Some alkynes undergo amination in the presence of gold(III) catalysts. Gold catalyses the alkylation of certain aromatic rings and the conversion of furans to phenols. For example, a mixture of acetonitrile and gold(III) chloride catalyses the alkylation of 2-methylfuran by methyl vinyl ketone at the 5-position:
The efficiency of this organogold reaction is noteworthy because both the furan and the ketone are sensitive to side reactions such as polymerisation under acidic conditions. In some cases where alkynes are present, phenols sometimes form (Ts is an abbreviation for tosyl):[13]
This reaction involves a rearrangement that gives a new aromatic ring.[14]
As a stoichiometric reagent, auric chloride reacts with benzene (and a variety of other arenes) under extremely mild conditions (reaction times of a few minutes at room temperature) to produce the dimeric phenylgold(III) dichloride:[15]
PhH + ½ Au2Cl6 → ½ [PhAuCl2]2 + HCl
References[]
- ^ Jump up to: a b c d e Egon Wiberg; Nils Wiberg; A. F. Holleman (2001). Inorganic Chemistry (101 ed.). Academic Press. pp. 1286–1287. ISBN 978-0-12-352651-9.
- ^ "Gold Chloride". American Elements. Retrieved July 22, 2019.
- ^ E. S. Clark; D. H. Templeton; C. H. MacGillavry (1958). "The crystal structure of gold(III) chloride". Acta Crystallogr. 11 (4): 284–288. doi:10.1107/S0365110X58000694. Retrieved 2010-05-21.
- ^ Buckley, Robbie W.; Healy, Peter C.; Loughlin, Wendy A. (1997). "Reduction of [NBu4][AuCl4] to [NBu4][AuCl2] with Sodium Acetylacetonate". Australian Journal of Chemistry. 50 (7): 775. doi:10.1071/C97029.
- ^ Block, B. P. (1953). "Gold Powder and Potassium Tetrabromoaurate(III)". Inorganic Syntheses. 4: 14–17. doi:10.1002/9780470132357.ch4.
- ^ N. N. Greenwood, A. Earnshaw, Chemistry of the Elements, 2nd ed., Butterworth-Heinemann, Oxford, UK, 1997
- ^ Handbook of Chemistry and Physics, 71st edition, CRC Press, Ann Arbor, Michigan, 1990
- ^ The Merck Index. An Encyclopaedia of Chemicals, Drugs and Biologicals. 14. Ed., 2006, p. 780, ISBN 978-0-911910-00-1.
- ^ H. Nechamkin, The Chemistry of the Elements, McGraw-Hill, New York, 1968
- ^ A. F. Wells, Structural Inorganic Chemistry, 5th ed., Oxford University Press, Oxford, UK, 1984
- ^ G. Dyker, An Eldorado for Homogeneous Catalysis?, in Organic Synthesis Highlights V, H.-G. Schmaltz, T. Wirth (eds.), pp 48–55, Wiley-VCH, Weinheim, 2003
- ^ Y. Fukuda; K. Utimoto (1991). "Effective transformation of unactivated alkynes into ketones or acetals with a gold(III) catalyst". J. Org. Chem. 56 (11): 3729. doi:10.1021/jo00011a058.
- ^ A. S. K. Hashmi; T. M. Frost; J. W. Bats (2000). "Highly Selective Gold-Catalyzed Arene Synthesis". J. Am. Chem. Soc. 122 (46): 11553. doi:10.1021/ja005570d.
- ^ A. Stephen; K. Hashmi; M. Rudolph; J. P. Weyrauch; M. Wölfle; W. Frey; J. W. Bats (2005). "Gold Catalysis: Proof of Arene Oxides as Intermediates in the Phenol Synthesis". Angewandte Chemie International Edition. 44 (18): 2798–801. doi:10.1002/anie.200462672. PMID 15806608.
- ^ Li, Zigang; Brouwer, Chad; He, Chuan (2008-08-01). "Gold-Catalyzed Organic Transformations". Chemical Reviews. 108 (8): 3239–3265. doi:10.1021/cr068434l. ISSN 0009-2665. PMID 18613729.
External links[]
 Media related to Gold trichloride at Wikimedia Commons
Media related to Gold trichloride at Wikimedia Commons
- Chlorides
- Metal halides
- Gold(III) compounds
- Deliquescent substances
- Photographic chemicals
- Gold–halogen compounds