Gold(III) fluoride

| |
| Names | |
|---|---|
| IUPAC name
Gold(III) fluoride
| |
| Other names
Gold trifluoride
Auric fluoride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
Chemical formula
|
AuF3 |
| Molar mass | 253.961779 g·mol−1 |
| Appearance | orange-yellow hexagonal crystals |
| Density | 6.75 g/cm3 |
| Melting point | sublimes above 300°C |
| Insoluble | |
Magnetic susceptibility (χ)
|
+74·10−6 cm3/mol |
| Structure | |
Crystal structure
|
Hexagonal, hP24 |
Space group
|
P6122, No. 178 |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
-363.3 kJ/mol |
| Related compounds | |
Other anions
|
Gold(III) chloride Gold(III) bromide |
Other cations
|
Silver fluoride Copper(II) fluoride Mercury(II) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Gold(III) fluoride, AuF3, is an orange solid that sublimes at 300 °C.[2] It is a powerful fluorinating agent.
Preparation[]
AuF3 can be prepared by reacting AuCl3 with F2 or BrF3.
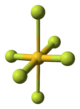
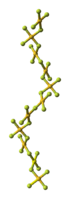
Structure[]
The crystal structure of AuF3 consists of spirals of square-planar AuF4 units.[3]
 |
 |
 |
 |
|
| AuF3 unit cell | neighbouring (AuF3)n helices | distorted octahedral coordination of gold by six fluorines | top-down view of an (AuF3)n helix | side view of an (AuF3)n helix |
References[]
- ^ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. 4–59. ISBN 0-8493-0594-2.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8., p. 1184.
- ^ F. W. B. Einstein; P. R. Rao; James Trotter; Neil Bartlett (1967). "The crystal structure of gold trifluoride". Journal of the Chemical Society A: Inorganic, Physical, Theoretical. 4: 478–482. doi:10.1039/J19670000478.
External links[]
 Media related to Gold trifluoride at Wikimedia Commons
Media related to Gold trifluoride at Wikimedia Commons
Categories:
- Fluorides
- Metal halides
- Gold(III) compounds
- Fluorinating agents
- Gold–halogen compounds
- Inorganic compound stubs