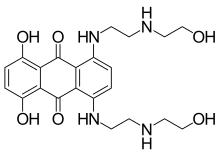
Mitoxantrone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Novantrone |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608019 |
| Routes of administration | Mainly intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | n/a |
| Protein binding | 78% |
| Metabolism | Hepatic (CYP2E1) |
| Elimination half-life | 75 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C22H28N4O6 |
| Molar mass | 444.488 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Mitoxantrone (INN, BAN, USAN; also known as Mitozantrone in Australia; trade name Novantrone) is an anthracenedione antineoplastic agent.
Uses[]
Mitoxantrone is used to treat certain types of cancer, mostly acute myeloid leukemia. It improves the survival rate of children suffering from acute lymphoblastic leukemia relapse.[1]
The combination of mitoxantrone and prednisone is approved as a second-line treatment for metastatic hormone-refractory prostate cancer. Until recently this combination was the first line of treatment; however, a combination of docetaxel and prednisone improves survival rates and lengthens the disease-free period.[2]
Mitoxantrone is also used to treat multiple sclerosis (MS), most notably the subset of the disease known as secondary-progressive MS. In the absence of a cure, mitoxantrone is effective in slowing the progression of secondary-progressive MS and extending the time between relapses in both relapsing-remitting MS and progressive-relapsing MS.[3]
Side effects[]
Mitoxantrone, as with other drugs in its class, may cause adverse reactions of varying severity, including nausea, vomiting, hair loss, heart damage and immunosuppression, possibly with delayed onset. Cardiomyopathy is a particularly concerning effect as it is irreversible; thus regular monitoring with echocardiograms or MUGA scans is recommended for patients.
Because of the risk of cardiomyopathy, mitoxantrone carries a limit on the cumulative lifetime dose (based on body surface area) in MS patients.[4]
Mechanism of action[]
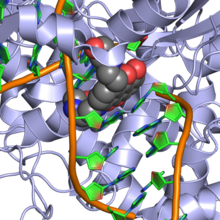
Mitoxantrone is a type II topoisomerase inhibitor; it disrupts DNA synthesis and DNA repair in both healthy cells and cancer cells by intercalation[6][7] between DNA bases. It is also classified as an antibiotic.[8]
See also[]
- Pixantrone, a mitoxantrone analogue under development
- Losoxantrone
References[]
- ^ Parker C, Waters R, Leighton C, Hancock J, Sutton R, Moorman AV, Ancliff P, Morgan M, Masurekar A, Goulden N, Green N, Révész T, Darbyshire P, Love S, Saha V (2010). "Effect of mitoxantrone on outcome of children with first relapse of acute lymphoblastic leukaemia (ALL R3): an open-label randomised trial". Lancet. 376 (9757): 2009–2017. doi:10.1016/S0140-6736(10)62002-8. PMC 3010035. PMID 21131038.
- ^ Katzung, Bertram G. (2006). "Cancer Chemotherapy". Basic and clinical pharmacology (10th ed.). New York: McGraw-Hill Medical Publishing Division. ISBN 0-07-145153-6. OCLC 157011367.
- ^ Fox E (2006). "Management of worsening multiple sclerosis with mitoxantrone: a review". Clin Ther. 28 (4): 461–74. doi:10.1016/j.clinthera.2006.04.013. PMID 16750460.
- ^ "Mitoxantrone Hydrochloride (marketed as Novantrone and generics) – Healthcare Professional Sheet text version". U.S. Food and Drug Administration. Retrieved 19 September 2014.
- ^ Wu, C. -C.; Li, Y. -C.; Wang, Y. -R.; Li, T. -K.; Chan, N. -L. (2013). "On the structural basis and design guidelines for type II topoisomerase-targeting anticancer drugs". Nucleic Acids Research. 41 (22): 10630–10640. doi:10.1093/nar/gkt828. PMC 3905874. PMID 24038465.
- ^ Mazerski J, Martelli S, Borowski E (1998). "The geometry of intercalation complex of antitumor mitoxantrone and ametantrone with DNA: molecular dynamics simulations". Acta Biochim. Pol. 45 (1): 1–11. doi:10.18388/abp.1998_4280. PMID 9701490.
- ^ Kapuscinski, J; Darzynkiewicz, Z (1985). "Interactions of antitumor agents Ametantrone and Mitoxantrone (Novatrone) with double-stranded DNA". Biochem Pharmacol. 34 (24): 4203–13. doi:10.1016/0006-2952(85)90275-8. PMID 4074383.
- ^ "Mitoxantrone".
External links[]
- "Mitoxantrone". Drug Information Portal. U.S. National Library of Medicine.
- Topoisomerase inhibitors
- IARC Group 2B carcinogens
- Primary alcohols
- Merck brands
- Aromatic amines
- Dihydroxyanthraquinones
- Hydroquinones
- DNA intercalaters
- 3-hydroxypropenals within hydroxyquinones