Sulfur
 | |||||||||||||||||||||||||||||||
| Sulfur | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alternative name | sulphur (British spelling) | ||||||||||||||||||||||||||||||
| Appearance | lemon yellow sintered microcrystals | ||||||||||||||||||||||||||||||
| Standard atomic weight Ar, std(S) | [32.059, 32.076] conventional: 32.06 | ||||||||||||||||||||||||||||||
| Sulfur in the periodic table | |||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||
| Atomic number (Z) | 16 | ||||||||||||||||||||||||||||||
| Group | group 16 (chalcogens) | ||||||||||||||||||||||||||||||
| Period | period 3 | ||||||||||||||||||||||||||||||
| Block | p-block | ||||||||||||||||||||||||||||||
| Electron configuration | [Ne] 3s2 3p4 | ||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 6 | ||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||
| Phase at STP | solid | ||||||||||||||||||||||||||||||
| Melting point | 388.36 K (115.21 °C, 239.38 °F) | ||||||||||||||||||||||||||||||
| Boiling point | 717.8 K (444.6 °C, 832.3 °F) | ||||||||||||||||||||||||||||||
| Density (near r.t.) | alpha: 2.07 g/cm3 beta: 1.96 g/cm3 gamma: 1.92 g/cm3 | ||||||||||||||||||||||||||||||
| when liquid (at m.p.) | 1.819 g/cm3 | ||||||||||||||||||||||||||||||
| Critical point | 1314 K, 20.7 MPa | ||||||||||||||||||||||||||||||
| Heat of fusion | mono: 1.727 kJ/mol | ||||||||||||||||||||||||||||||
| Heat of vaporization | mono: 45 kJ/mol | ||||||||||||||||||||||||||||||
| Molar heat capacity | 22.75 J/(mol·K) | ||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||
| Oxidation states | −2, −1, 0, +1, +2, +3, +4, +5, +6 (a strongly acidic oxide) | ||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 2.58 | ||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||
| Covalent radius | 105±3 pm | ||||||||||||||||||||||||||||||
| Van der Waals radius | 180 pm | ||||||||||||||||||||||||||||||
| Other properties | |||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||
| Crystal structure | orthorhombic | ||||||||||||||||||||||||||||||
| Thermal conductivity | 0.205 W/(m⋅K) (amorphous) | ||||||||||||||||||||||||||||||
| Electrical resistivity | 2×1015 Ω⋅m (at 20 °C) (amorphous) | ||||||||||||||||||||||||||||||
| Magnetic ordering | diamagnetic[1] | ||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | (α) −15.5×10−6 cm3/mol (298 K)[2] | ||||||||||||||||||||||||||||||
| Bulk modulus | 7.7 GPa | ||||||||||||||||||||||||||||||
| Mohs hardness | 2.0 | ||||||||||||||||||||||||||||||
| CAS Number | 7704-34-9 | ||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||
| Discovery | Chinese[3] (before 2000 BCE) | ||||||||||||||||||||||||||||||
| Recognized as an element by | Antoine Lavoisier (1777) | ||||||||||||||||||||||||||||||
| Main isotopes of sulfur | |||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||
Sulfur (in British English: sulphur) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature.
Sulfur is the tenth most common element by mass in the universe, and the fifth most common on Earth. Though sometimes found in pure, native form, sulfur on Earth usually occurs as sulfide and sulfate minerals. Being abundant in native form, sulfur was known in ancient times, being mentioned for its uses in ancient India, ancient Greece, China, and Egypt. Historically and in literature sulfur is also called brimstone,[4] which means "burning stone".[5] Today, almost all elemental sulfur is produced as a byproduct of removing sulfur-containing contaminants from natural gas and petroleum. The greatest commercial use of the element is the production of sulfuric acid for sulfate and phosphate fertilizers, and other chemical processes. Sulfur is used in matches, insecticides, and fungicides. Many sulfur compounds are odoriferous, and the smells of odorized natural gas, skunk scent, grapefruit, and garlic are due to organosulfur compounds. Hydrogen sulfide gives the characteristic odor to rotting eggs and other biological processes.
Sulfur is an essential element for all life, but almost always in the form of organosulfur compounds or metal sulfides. Three amino acids (cysteine, cystine, and methionine) and two vitamins (biotin and thiamine) are organosulfur compounds. Many cofactors also contain sulfur, including glutathione, thioredoxin, and iron–sulfur proteins. Disulfides, S–S bonds, confer mechanical strength and insolubility of the protein keratin, found in outer skin, hair, and feathers. Sulfur is one of the core chemical elements needed for biochemical functioning and is an elemental macronutrient for all living organisms.
Characteristics[]

Physical properties[]
Sulfur forms several polyatomic molecules. The best-known allotrope is octasulfur, cyclo-S8. The point group of cyclo-S8 is D4d and its dipole moment is 0 D.[6] Octasulfur is a soft, bright-yellow solid that is odorless, but impure samples have an odor similar to that of matches.[7] It melts at 115.21 °C (239.38 °F), boils at 444.6 °C (832.3 °F) and sublimates easily.[4] At 95.2 °C (203.4 °F), below its melting temperature, cyclo-octasulfur changes from α-octasulfur to the β-polymorph.[8] The structure of the S8 ring is virtually unchanged by this phase change, which affects the intermolecular interactions. Between its melting and boiling temperatures, octasulfur changes its allotrope again, turning from β-octasulfur to γ-sulfur, again accompanied by a lower density but increased viscosity due to the formation of polymers.[8] At higher temperatures, the viscosity decreases as depolymerization occurs. Molten sulfur assumes a dark red color above 200 °C (392 °F). The density of sulfur is about 2 g/cm3, depending on the allotrope; all of the stable allotropes are excellent electrical insulators.
Chemical properties[]
Sulfur burns with a blue flame with formation of sulfur dioxide, which has a suffocating and irritating odor. Sulfur is insoluble in water but soluble in carbon disulfide and, to a lesser extent, in other nonpolar organic solvents, such as benzene and toluene. The first and second ionization energies of sulfur are 999.6 and 2252 kJ/mol, respectively. Despite such figures, the +2 oxidation state is rare, with +4 and +6 being more common. The fourth and sixth ionization energies are 4556 and 8495.8 kJ/mol, the magnitude of the figures caused by electron transfer between orbitals; these states are only stable with strong oxidants such as fluorine, oxygen, and chlorine.[citation needed] Sulfur reacts with nearly all other elements with the exception of the noble gases, even with the notoriously unreactive metal iridium (yielding iridium disulfide).[9] Some of those reactions need elevated temperatures.[10]
Isotopes[]
Sulfur has 23 known isotopes, four of which are stable: 32S (94.99%±0.26%), 33S (0.75%±0.02%), 34S (4.25%±0.24%), and 36S (0.01%±0.01%).[11][12] Other than 35S, with a half-life of 87 days and formed in cosmic ray spallation of 40Ar, the radioactive isotopes of sulfur have half-lives less than 3 hours.
When sulfide minerals are precipitated, isotopic equilibration among solids and liquid may cause small differences in the δ34S values of co-genetic minerals. The differences between minerals can be used to estimate the temperature of equilibration. The δ13C and δ34S of coexisting carbonate minerals and sulfides can be used to determine the pH and oxygen fugacity of the ore-bearing fluid during ore formation.
In most forest ecosystems, sulfate is derived mostly from the atmosphere; weathering of ore minerals and evaporites contribute some sulfur. Sulfur with a distinctive isotopic composition has been used to identify pollution sources, and enriched sulfur has been added as a tracer in hydrologic studies. Differences in the natural abundances can be used in systems where there is sufficient variation in the 34S of ecosystem components. Rocky Mountain lakes thought to be dominated by atmospheric sources of sulfate have been found to have characteristic 34S values from lakes believed to be dominated by watershed sources of sulfate.
Natural occurrence[]



32S is created inside massive stars, at a depth where the temperature exceeds 2.5×109 K, by the fusion of one nucleus of silicon plus one nucleus of helium.[13] As this nuclear reaction is part of the alpha process that produces elements in abundance, sulfur is the 10th most common element in the universe.
Sulfur, usually as sulfide, is present in many types of meteorites. Ordinary chondrites contain on average 2.1% sulfur, and carbonaceous chondrites may contain as much as 6.6%. It is normally present as troilite (FeS), but there are exceptions, with carbonaceous chondrites containing free sulfur, sulfates and other sulfur compounds.[14] The distinctive colors of Jupiter's volcanic moon Io are attributed to various forms of molten, solid, and gaseous sulfur.[15]
It is the fifth most common element by mass in the Earth. Elemental sulfur can be found near hot springs and volcanic regions in many parts of the world, especially along the Pacific Ring of Fire; such volcanic deposits are currently mined in Indonesia, Chile, and Japan. These deposits are polycrystalline, with the largest documented single crystal measuring 22×16×11 cm.[16] Historically, Sicily was a major source of sulfur in the Industrial Revolution.[17] Lakes of molten sulfur up to ~200 m in diameter have been found on the sea floor, associated with submarine volcanoes, at depths where the boiling point of water is higher than the melting point of sulfur.[18]
Native sulfur is synthesised by anaerobic bacteria acting on sulfate minerals such as gypsum in salt domes.[19][20] Significant deposits in salt domes occur along the coast of the Gulf of Mexico, and in evaporites in eastern Europe and western Asia. Native sulfur may be produced by geological processes alone. Fossil-based sulfur deposits from salt domes were once the basis for commercial production in the United States, Russia, Turkmenistan, and Ukraine.[21] Currently, commercial production is still carried out in the Osiek mine in Poland. Such sources are now of secondary commercial importance, and most are no longer worked.
Common naturally occurring sulfur compounds include the sulfide minerals, such as pyrite (iron sulfide), cinnabar (mercury sulfide), galena (lead sulfide), sphalerite (zinc sulfide), and stibnite (antimony sulfide); and the sulfate minerals, such as gypsum (calcium sulfate), alunite (potassium aluminium sulfate), and barite (barium sulfate). On Earth, just as upon Jupiter's moon Io, elemental sulfur occurs naturally in volcanic emissions, including emissions from hydrothermal vents.
Compounds[]
Common oxidation states of sulfur range from −2 to +6. Sulfur forms stable compounds with all elements except the noble gases.
Allotropes[]

Sulfur forms over 30 solid allotropes, more than any other element.[22] Besides S8, several other rings are known.[23] Removing one atom from the crown gives S7, which is more of a deep yellow than the S8. HPLC analysis of "elemental sulfur" reveals an equilibrium mixture of mainly S8, but with S7 and small amounts of S6.[24] Larger rings have been prepared, including S12 and S18.[25][26]
Amorphous or "plastic" sulfur is produced by rapid cooling of molten sulfur—for example, by pouring it into cold water. X-ray crystallography studies show that the amorphous form may have a helical structure with eight atoms per turn. The long coiled polymeric molecules make the brownish substance elastic, and in bulk this form has the feel of crude rubber. This form is metastable at room temperature and gradually reverts to crystalline molecular allotrope, which is no longer elastic. This process happens within a matter of hours to days, but can be rapidly catalyzed.
Polycations and polyanions[]

Sulfur polycations, S82+, S42+ and S162+ are produced when sulfur is reacted with mild oxidising agents in a strongly acidic solution.[27] The colored solutions produced by dissolving sulfur in oleum were first reported as early as 1804 by C.F. Bucholz, but the cause of the color and the structure of the polycations involved was only determined in the late 1960s. S82+ is deep blue, S42+ is yellow and S162+ is red.[8]
The radical anion S3− gives the blue color of the mineral lapis lazuli.

Sulfides[]
Treatment of sulfur with hydrogen gives hydrogen sulfide. When dissolved in water, hydrogen sulfide is mildly acidic:[4]
- H2S ⇌ HS− + H+
Hydrogen sulfide gas and the hydrosulfide anion are extremely toxic to mammals, due to their inhibition of the oxygen-carrying capacity of hemoglobin and certain cytochromes in a manner analogous to cyanide and azide (see below, under precautions).
Reduction of elemental sulfur gives polysulfides, which consist of chains of sulfur atoms terminated with S− centers:
- 2 Na + S8 → Na2S8
This reaction highlights a distinctive property of sulfur: its ability to catenate (bind to itself by formation of chains). Protonation of these polysulfide anions produces the polysulfanes, H2Sx where x= 2, 3, and 4.[29] Ultimately, reduction of sulfur produces sulfide salts:
- 16 Na + S8 → 8 Na2S
The interconversion of these species is exploited in the sodium–sulfur battery.
Oxides, oxoacids, and oxoanions[]
The principal sulfur oxides are obtained by burning sulfur:
- S + O2 → SO2 (sulfur dioxide)
- 2 SO2 + O2 → 2 SO3 (sulfur trioxide)
Multiple sulfur oxides are known; the sulfur-rich oxides include sulfur monoxide, disulfur monoxide, disulfur dioxides, and higher oxides containing peroxo groups.
Sulfur forms sulfur oxoacids, some of which cannot be isolated and are only known through the salts. Sulfur dioxide and sulfites (SO2−
3) are related to the unstable sulfurous acid (H2SO3). Sulfur trioxide and sulfates (SO2−
4) are related to sulfuric acid (H2SO4). Sulfuric acid and SO3 combine to give oleum, a solution of pyrosulfuric acid (H2S2O7) in sulfuric acid.
Thiosulfate salts (S
2O2−
3), sometimes referred as "hyposulfites", used in photographic fixing (hypo) and as reducing agents, feature sulfur in two oxidation states. Sodium dithionite (Na
2S
2O
4), contains the more highly reducing dithionite anion (S
2O2−
4).
Halides and oxyhalides[]
Several sulfur halides are important to modern industry. Sulfur hexafluoride is a dense gas used as an insulator gas in high voltage transformers; it is also a nonreactive and nontoxic propellant for pressurized containers. Sulfur tetrafluoride is a rarely-used organic reagent that is highly toxic.[30] Sulfur dichloride and disulfur dichloride are important industrial chemicals. Sulfuryl chloride and chlorosulfuric acid are derivatives of sulfuric acid; thionyl chloride (SOCl2) is a common reagent in organic synthesis.[31]
Pnictides[]
An important S–N compound is the cage tetrasulfur tetranitride (S
4N
4). Heating this compound gives polymeric sulfur nitride (SN
x), which has metallic properties even though it does not contain any metal atoms. Thiocyanates contain the SCN−
group. Oxidation of thiocyanate gives thiocyanogen, (SCN)
2 with the connectivity NCS−SCN. Phosphorus sulfides are numerous, the most important commercially being the cages P
4S
10 and P
4S
3.[32][33]
Metal sulfides[]
The principal ores of copper, zinc, nickel, cobalt, molybdenum, and other metals are sulfides. These materials tend to be dark-colored semiconductors that are not readily attacked by water or even many acids. They are formed, both geochemically and in the laboratory, by the reaction of hydrogen sulfide with metal salts. The mineral galena (PbS) was the first demonstrated semiconductor and was used as a signal rectifier in the cat's whiskers of early crystal radios. The iron sulfide called pyrite, the so-called "fool's gold", has the formula FeS2.[34] Processing these ores, usually by roasting, is costly and environmentally hazardous. Sulfur corrodes many metals through tarnishing.
Organic compounds[]
- Illustrative organosulfur compounds
Allicin, a chemical compound in garlic

(R)-cysteine, an amino acid containing a thiol group
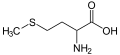
Methionine, an amino acid containing a thioether

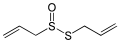
Diphenyl disulfide, a representative disulfide

Perfluorooctanesulfonic acid, a surfactant
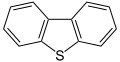
Dibenzothiophene, a component of crude oil
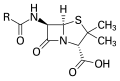
Penicillin, an antibiotic where "R" is the variable group
Some of the main classes of sulfur-containing organic compounds include the following:[35]
- Thiols or mercaptans (so called because they capture mercury as chelators) are the sulfur analogs of alcohols; treatment of thiols with base gives thiolate ions.
- Thioethers are the sulfur analogs of ethers.
- Sulfonium ions have three groups attached to a cationic sulfur center. Dimethylsulfoniopropionate (DMSP) is one such compound, important in the marine organic sulfur cycle.
- Sulfoxides and sulfones are thioethers with one and two oxygen atoms attached to the sulfur atom, respectively. The simplest sulfoxide, dimethyl sulfoxide, is a common solvent; a common sulfone is sulfolane.
- Sulfonic acids are used in many detergents.
Compounds with carbon–sulfur multiple bonds are uncommon, an exception being carbon disulfide, a volatile colorless liquid that is structurally similar to carbon dioxide. It is used as a reagent to make the polymer rayon and many organosulfur compounds. Unlike carbon monoxide, carbon monosulfide is stable only as an extremely dilute gas, found between solar systems.[36]
Organosulfur compounds are responsible for some of the unpleasant odors of decaying organic matter. They are widely known as the odorant in domestic natural gas, garlic odor, and skunk spray. Not all organic sulfur compounds smell unpleasant at all concentrations: the sulfur-containing monoterpenoid (grapefruit mercaptan) in small concentrations is the characteristic scent of grapefruit, but has a generic thiol odor at larger concentrations. Sulfur mustard, a potent vesicant, was used in World War I as a disabling agent.[37]
Sulfur–sulfur bonds are a structural component used to stiffen rubber, similar to the disulfide bridges that rigidify proteins (see biological below). In the most common type of industrial "curing" or hardening and strengthening of natural rubber, elemental sulfur is heated with the rubber to the point that chemical reactions form disulfide bridges between isoprene units of the polymer. This process, patented in 1843, made rubber a major industrial product, especially in automobile tires. Because of the heat and sulfur, the process was named vulcanization, after the Roman god of the forge and volcanism.
History[]
Antiquity[]

Being abundantly available in native form, sulfur was known in ancient times and is referred to in the Torah (Genesis). English translations of the Bible commonly referred to burning sulfur as "brimstone", giving rise to the term "fire-and-brimstone" sermons, in which listeners are reminded of the fate of eternal damnation that await the unbelieving and unrepentant. It is from this part of the Bible[citation needed] that Hell is implied to "smell of sulfur" (likely due to its association with volcanic activity). According to the Ebers Papyrus, a sulfur ointment was used in ancient Egypt to treat granular eyelids. Sulfur was used for fumigation in preclassical Greece;[38] this is mentioned in the Odyssey.[39] Pliny the Elder discusses sulfur in book 35 of his Natural History, saying that its best-known source is the island of Melos. He mentions its use for fumigation, medicine, and bleaching cloth.[40]
A natural form of sulfur known as shiliuhuang (石硫黄) was known in China since the 6th century BC and found in Hanzhong.[41] By the 3rd century, the Chinese discovered that sulfur could be extracted from pyrite.[41] Chinese Daoists were interested in sulfur's flammability and its reactivity with certain metals, yet its earliest practical uses were found in traditional Chinese medicine.[41] A Song dynasty military treatise of 1044 AD described various formulas for Chinese black powder, which is a mixture of potassium nitrate (KNO
3), charcoal, and sulfur. It remains an ingredient of black gunpowder.
Indian alchemists, practitioners of "the science of chemicals" (sanskrit rasaśāstra, रसशास्त्र), wrote extensively about the use of sulfur in alchemical operations with mercury, from the eighth century AD onwards.[42] In the rasaśāstra tradition, sulfur is called "the smelly" (sanskrit gandhaka, गन्धक).
Early European alchemists gave sulfur a unique alchemical symbol, a triangle at the top of a cross (








