Setoperone
| |
| Names | |
|---|---|
| Preferred IUPAC name
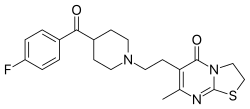
6-{2-[4-(4-Fluorobenzoyl)piperidin-1-yl]ethyl}-7-methyl-2,3-dihydro-5H-[1,3]thiazolo[3,2-a]pyrimidin-5-one | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| C21H24FN3O2S | |
| Molar mass | 401.50 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Setoperone is a compound that is a ligand to the 5-HT2A receptor. It can be radiolabeled with the radioisotope fluorine-18 and used as a radioligand with positron emission tomography (PET). Several research studies have used the radiolabeled setoperone in neuroimaging for the studying neuropsychiatric disorders, such as depression[1] or schizophrenia.[2]
References[]
- ^ Jeffrey H. Meyer, Shitij Kapur, , Jean DaSilva, Beata Owczarek, Gregory M. Brown, Alan A. Wilson and Sidney H. Kennedy (July 1, 1999). "Prefrontal Cortex 5-HT2 Receptors in Depression: An [18F]Setoperone PET Imaging Study". American Journal of Psychiatry. 156 (7): 1029–1034. doi:10.1176/ajp.156.7.1029 (inactive 31 May 2021). PMID 10401447.CS1 maint: multiple names: authors list (link) CS1 maint: DOI inactive as of May 2021 (link)
- ^ Ralph Lewis, Shitij Kapur, Corey Jones, Jean DaSilva, Gregory M. Brown, Alan A. Wilson, and Robert B. Zipursky (January 1, 1999). "Serotonin 5-HT2 Receptors in Schizophrenia: A PET Study Using [18F]Setoperone in Neuroleptic-Naive Patients and Normal Subjects". American Journal of Psychiatry. 156 (1): 72–78. doi:10.1176/ajp.156.1.72. PMID 9892300.CS1 maint: multiple names: authors list (link)
Categories:
- 5-HT2A antagonists
- Abandoned drugs
- Aromatic ketones
- Fluoroarenes
- Piperidines
- Receptor modulators
- Thiazolopyrimidines
- Nervous system drug stubs