Disulfur dibromide
| |
| |
| Names | |
|---|---|
| IUPAC name
bromosulfanyl thiohypobromite
| |
| Other names
Dibromodisulfane
Bromosulfanyl thiohypobromite | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.032.821 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Br2S2 | |
| Molar mass | 223.940 g mol−1 |
| Appearance | orange/yellow liquid |
| Density | 2.703 g/cm3 |
| Boiling point | 46–48 °C (115–118 °F; 319–321 K) 0.1 mm Hg |
| Structure | |
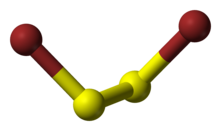
| C2, gauche | |
| Hazards | |
| Safety data sheet (SDS) | ICSC 1661 |
| GHS labelling: | |
  
| |
Signal word
|
Danger |
| Related compounds | |
Related
|
Sulfur dibromide Thionyl bromide |
Related compounds
|
Disulfur difluoride Disulfur dichloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Disulfur dibromide is the inorganic compound with the formula S2Br2. It is a yellow-brown liquid that fumes in air. It is prepared by direct combination of the elements and purified by vacuum distillation.[1] The compound has no particular application, unlike the related sulfur compound disulfur dichloride.
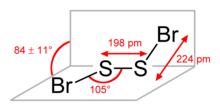
The molecular structure is akin to that of disulfur dichloride (S2Cl2). According to electron diffraction measurements, the BrSSBr dihedral angle is 84° and the Br-S-S angle is 107°. The S-S distance is 1.980 Å, ca. 0.050 Å shorter than for S2Cl2.[2]
References[]
- ^ F. Fehér (1963). "Dibromodisulfane". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. 1. NY, NY: Academic Press. pp. 377–378.
- ^ Zysman-Colman, Eli; Harpp, David (2004). "Comparison of the Structural Properties of Compounds Containing the XSSX Moiety (X = H, Me, R, Cl, Br, F, OR)". Journal of Sulfur Chemistry. 25: 291-316. doi:10.1080/17415990410001710163. S2CID 95468251.
Categories:
- Bromides
- Disulfides
- Sulfur halides