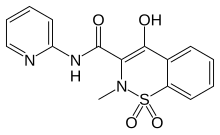
Oxicam
Oxicam is a class of nonsteroidal anti-inflammatory drugs (NSAIDs) that bind closely to plasma proteins.[1] Most oxicams are unselective inhibitors of the cyclooxygenase (COX) enzymes. The exception is meloxicam with a slight (10:1) preference for COX-2, which, however, is only clinically relevant at low doses.[2]
Examples include:
- Ampiroxicam
- Piroxicam
- Tenoxicam
- Droxicam
- Lornoxicam
- Meloxicam
- Isoxicam
Chemistry[]
The physico-chemical characteristics of these molecules vary greatly depending upon the environment.[3]
In contrast to most other NSAIDs, oxicams are not carboxylic acids. They are tautomeric and can exist as a number of tautomers (keto-enol tautomerism), here exemplified by piroxicam:[4]

Side Effects[]
The oxicams are associated with drug-related erythema multiforme (EM), Stevens–Johnson syndrome, and toxic epidermal necrolysis (TEN). This association is one of the reasons Oxicams are not regularly prescribed.
References[]
- ^ Olkkola KT, Brunetto AV, Mattila MJ (1994). "Pharmacokinetics of oxicam nonsteroidal anti-inflammatory agents". Clinical Pharmacokinetics. 26 (2): 107–20. doi:10.2165/00003088-199426020-00004. PMID 8162655.
- ^ Mutschler, Ernst; Gerd Geisslinger; Heyo K. Kroemer; Monika Schäfer-Korting (2001). Arzneimittelwirkungen (in German) (8 ed.). Stuttgart: Wissenschaftliche Verlagsgesellschaft. p. 233. ISBN 3-8047-1763-2.
- ^ Banerjee R, Chakraborty H, Sarkar M (2003). "Photophysical studies of oxicam group of NSAIDs: piroxicam, meloxicam and tenoxicam". Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 59 (6): 1213–22. Bibcode:2003AcSpA..59.1213B. doi:10.1016/S1386-1425(02)00300-1. PMID 12659890.
- ^ Ivanova D, Deneva V, Nedeltcheva D, Kamounah FS, Gergov G, Hansen PE, Kawauchi S, Antonov L (2015). "Tautomeric transformations of piroxicam in solution: a combined experimental and theoretical study". RSC Advances. 5 (40): 31852–31860. doi:10.1039/c5ra03653d.
- Nonsteroidal anti-inflammatory drugs
- Sultams
- Musculoskeletal system drug stubs