From Wikipedia, the free encyclopedia
Icilin
Names
Preferred IUPAC name
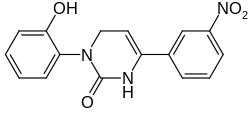
3-(2-Hydroxyphenyl)-6-(3-nitrophenyl)-3,4-dihydropyrimidin-2(1H )-one
Other names
1-(2-Hydroxyphenyl)-4-(3-nitrophenyl)-3,6-dihydropyrimidin-2-one
Identifiers
3D model (JSmol )
ChemSpider
ECHA InfoCard 100.164.593
IUPHAR/BPS
UNII
InChI=1S/C16H13N3O4/c20-15-7-2-1-6-14(15)18-9-8-13(17-16(18)21)11-4-3-5-12(10-11)19(22)23/h1-8,10,20H,9H2,(H,17,21)
N Key: RCEFMOGVOYEGJN-UHFFFAOYSA-N
N InChI=1/C16H13N3O4/c20-15-7-2-1-6-14(15)18-9-8-13(17-16(18)21)11-4-3-5-12(10-11)19(22)23/h1-8,10,20H,9H2,(H,17,21)
Key: RCEFMOGVOYEGJN-UHFFFAOYAQ
O=C3N(c1c(O)cccc1)C/C=C(/c2cccc([N+]([O-])=O)c2)N3
Properties
C16 H13 N3 O4
Molar mass
311.29 g/mol
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
N what is Y N
Infobox references
Chemical compound
Icilin (AG-3-5 ) is a synthetic super-agonist of the transient receptor potential M8 (TRPM8 ) ion channel . Although structurally not related with menthol , it produces an extreme sensation of cold, both in humans and animals. It is almost 200 times more potent than menthol, and 2.5 times more efficacious.[1] [2] pruritus in an experimental model of itch.[3] TRP channels , although despite its high potency it is less selective for TRPM8 over other related ion channels than newer compounds such as WS-12 .
References [ ]
^ Wei ET, Seid DA (1983). "AG-3-5: a chemical producing sensations of cold". J. Pharm. Pharmacol . 35 (2): 110–2. doi :10.1111/j.2042-7158.1983.tb04279.x . PMID 6131976 . S2CID 42844929 . ^ Andersson DA, Chase HW, Bevan S (2004). "TRPM8 activation by menthol, icilin, and cold is differentially modulated by intracellular pH" . J. Neurosci . 24 (23): 5364–9. doi :10.1523/JNEUROSCI.0890-04.2004 PMC 6729305 PMID 15190109 . ^ Biró, T; Ko, MC; Bromm, B; Wei, ET; Bigliardi, P; Siebenhaar, F; Hashizume, H; Misery, L; et al. (2005). "How best to fight that nasty itch - from new insights into the neuroimmunological, neuroendocrine, and neurophysiological bases of pruritus to novel therapeutic approaches" . Experimental Dermatology . 14 (3): 225–40. doi :10.1111/j.0906-6705.2005.0321a.x PMID 15740597 . S2CID 23665244 .
External links [ ]
TRPA
Activators
4-Hydroxynonenal 4-Oxo-2-nonenal 4,5-EET 12S-HpETE 15-Deoxy-Δ12,14 -prostaglandin J2 α-Sanshool (ginger , Sichuan and melegueta peppers )Acrolein Allicin (garlic )Allyl isothiocyanate (mustard , radish , horseradish , wasabi )AM404 ASP-7663 Bradykinin Cannabichromene (cannabis )Cannabidiol (cannabis )Cannabigerol (cannabis )Cinnamaldehyde (cinnamon )CR gas (dibenzoxazepine; DBO) CS gas (2-chlorobenzal malononitrile) Cuminaldehyde (cumin )Curcumin (turmeric ) (celery )
Diallyl disulfide Dicentrine (Lindera Formalin Gingerols (ginger )Hepoxilin A3 Hepoxilin B3 Hydrogen peroxide Icilin Isothiocyanate JT-010 (celery , Angelica acutiloba
Linalool (Sichuan pepper , thyme )Methylglyoxal Methyl salicylate (wintergreen )N-Methylmaleimide Nicotine (tobacco )Oleocanthal (olive oil )Paclitaxel (Pacific yew )Paracetamol (acetaminophen) PF-4840154 Phenacyl chloride Polygodial (Dorrigo pepper )Shogaols (ginger , Sichuan and melegueta peppers )Tear gases Tetrahydrocannabinol (cannabis )Tetrahydrocannabiorcol Thiopropanal S-oxide (onion )Umbellulone (Umbellularia californica )WIN 55,212-2 Blockers
TRPC
TRPM
TRPML
TRPP
TRPV
Activators
2-APB 5',6'-EET 9-HODE 9-oxoODE 12S-HETE 12S-HpETE 13-HODE 13-oxoODE 20-HETE α-Sanshool (ginger , Sichuan and melegueta peppers )Allicin (garlic )AM404 Anandamide (Andrographis paniculata
Camphor (camphor laurel , rosemary , camphorweed , African blue basil , camphor basil )Cannabidiol (cannabis )Cannabidivarin (cannabis )Capsaicin (chili pepper )Carvacrol (oregano , thyme , pepperwort , wild bergamot , others)DHEA Diacyl glycerol Dihydrocapsaicin (chili pepper )Estradiol Eugenol (basil , clove )Evodiamine (Euodia ruticarpa Gingerols (ginger )GSK1016790A Heat Hepoxilin A3 Hepoxilin B3 Homocapsaicin (chili pepper )Homodihydrocapsaicin (chili pepper )Incensole (incense )Lysophosphatidic acid Low pH (acidic conditions)
Menthol (mint )N-Arachidonoyl dopamine N-Oleoylethanolamide Nonivamide (PAVA) (PAVA spray )Nordihydrocapsaicin (chili pepper )Paclitaxel (Pacific yew )Paracetamol (acetaminophen) Phenylacetylrinvanil Phorbol esters (e.g., )Piperine (black pepper , long pepper )Polygodial (Dorrigo pepper )Probenecid Protons RhTx (Ruta graveolens
Resiniferatoxin (RTX) (Euphorbia resinifera /pooissonii Shogaols (ginger , Sichuan and melegueta peppers )Tetrahydrocannabivarin (cannabis )Thymol (thyme , oregano )Tinyatoxin (Euphorbia resinifera /pooissonii Tramadol Vanillin (vanilla )Zucapsaicin Blockers
See also: Receptor/signaling modulators • Ion channel modulators
Categories :
Phenols Nitrobenzenes Ureas Pyrimidones Cooling flavors Organic compound stubs Hidden categories:
Articles without EBI source Articles without KEGG source Chembox CAS registry number linked Articles with changed ChemSpider identifier ECHA InfoCard ID from Wikidata Articles with changed InChI identifier Pages using collapsible list with both background and text-align in titlestyle Articles containing unverified chemical infoboxes Chembox image size set Articles with short description Short description matches Wikidata All stub articles