Arbaclofen placarbil
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.221.150 |
| Chemical and physical data | |
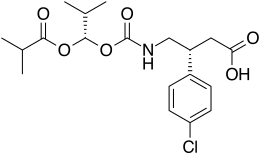
| Formula | C19H26ClNO6 |
| Molar mass | 399.87 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Arbaclofen placarbil (/ɑːrˈbækloʊfɛn pləˈkɑːrbɪl/ ar-BAK-loh-fen plə-KAR-bil, also known as XP19986) is a prodrug of R-baclofen. Arbaclofen placarbil possesses more favorable pharmacokinetic profile than baclofen, with less fluctuations in plasma drug levels. It was being developed as a potential treatment for patients with GERD and spasticity due to multiple sclerosis; however, in May 2013 XenoPort announced the termination of development because of unsuccessful results in phase III clinical trials.[1]
It is being developed as an addiction medicine to treat alcoholism. [2] It is also studied as a potential therapeutic for some autistic subjects.[3]
See also[]
- Gabapentin enacarbil
- Lesogaberan
References[]
- ^ "XenoPort Reports Top-Line Results of Phase 3 Trial of Arbaclofen Placarbil for Spasticity in Multiple Sclerosis Patients". XenoPort, Inc. May 20, 2013. Archived from the original on 2013-12-02. Retrieved 2013-06-03.
- ^ Anderson E (3 July 2020). "Pill that replaces alcohol aims to end 'glass of wine' craving". The i newspaper. Associated Newspapers Limited.
- ^ "GABAB receptor modulation of visual sensory processing in adults with and without autism spectrum disorder". Science translational medicine. 2022-01-05.
Categories:
- Drugs not assigned an ATC code
- Abandoned drugs
- Amino acids
- Calcium channel blockers
- Chloroarenes
- GABA analogues
- GABAB receptor agonists
- Prodrugs
- Gastrointestinal system drug stubs
- Musculoskeletal system drug stubs