Oxygen difluoride

| |
| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.087 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
Chemical formula
|
OF 2 |
| Molar mass | 53.9962 g/mol |
| Appearance | colorless gas, pale yellow liquid when condensed |
| Odor | peculiar, foul |
| Density |
|
| Melting point | −223.8 °C (−370.8 °F; 49.3 K) |
| Boiling point | −144.75 °C (−228.55 °F; 128.40 K) |
| hydrolyzes[1] | |
| Vapor pressure | 48.9 atm (at −58.0 °C or −72.4 °F or 215.2 K[a]) |
| Thermochemistry | |
Heat capacity (C)
|
43.3 J/mol K |
Std molar
entropy (S |
246.98 J/mol K |
Std enthalpy of
formation (ΔfH⦵298) |
24.5 kJ mol−1 |
Gibbs free energy (ΔfG˚)
|
42.5 kJ/mol |
| Hazards | |
EU classification (DSD) (outdated)
|
|
| NFPA 704 (fire diamond) | 
4
0
3 OX |
| Lethal dose or concentration (LD, LC): | |
LC50 (median concentration)
|
|
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.05 ppm (0.1 mg/m3)[2] |
REL (Recommended)
|
C 0.05 ppm (0.1 mg/m3)[2] |
IDLH (Immediate danger)
|
0.5 ppm[2] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
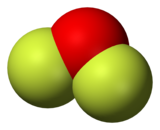
Oxygen difluoride is a chemical compound with the formula OF
2. As predicted by VSEPR theory, the molecule adopts a "bent" molecular geometry similar to that of water. However, it has very different properties, being a strong oxidizer.
Preparation[]
Oxygen difluoride was first reported in 1929; it was obtained by the electrolysis of molten potassium fluoride and hydrofluoric acid containing small quantities of water.[4][5] The modern preparation entails the reaction of fluorine with a dilute aqueous solution of sodium hydroxide, with sodium fluoride as a side-product:
- 2 F
2 + 2 NaOH → OF
2 + 2 NaF + H
2O
Reactions[]
Its powerful oxidizing properties are suggested by the oxidation number of +2 for the oxygen atom instead of its normal −2. Above 200 °C, OF
2 decomposes to oxygen and fluorine via a radical mechanism.
OF
2 reacts with many metals to yield oxides and fluorides. Nonmetals also react: phosphorus reacts with OF
2 to form PF
5 and POF
3; sulfur gives SO
2 and SF
4; and unusually for a noble gas, xenon reacts (at elevated temperatures) yielding XeF
4 and xenon oxyfluorides.
Oxygen difluoride reacts very slowly with water to form hydrofluoric acid:
- OF
2
(aq) + H
2O
(l) → 2 HF
(aq) + O
2
(g)
It can oxidize sulphur dioxide to sulfur trioxide and elemental fluorine:
- OF
2 + SO
2 → SO
3 + F
2
However, in the presence of UV radiation the products are sulfuryl fluoride (SO
2F
2) and pyrosulfuryl fluoride (S
2O
5F
2):
- OF
2 + 2 SO
2 → S
2O
5F
2
Safety[]
This section needs expansion. You can help by . (August 2018) |
Oxygen difluoride is considered an unsafe gas due to its oxidizing properties. Hydrofluoric acid produced by the hydrolysis of OF
2 with water is highly corrosive and toxic, capable of causing necrosis, leaching calcium from the bones and causing cardiovascular damage, among a host of other insidious effects.
Popular culture[]
In Robert L. Forward's science fiction novel Camelot 30K, oxygen difluoride was used as a biochemical solvent by fictional life forms living in the solar system's Kuiper belt. While OF
2 would be a solid at 30 K, the fictional alien lifeforms were described as endothermic, maintaining elevated body temperatures and liquid OF
2 blood by radiothermal heating.
Notes[]
- ^ This is its critical temperature, which is below ordinary room temperature.
References[]
- ^ "difluorine monoxide; oxygen difluoride, physical properties, suppliers, CAS, MSDS, structure, Molecular Formula, Molecular Weight, Solubility, boiling point, melting point". www.chemyq.com.
- ^ Jump up to: a b c NIOSH Pocket Guide to Chemical Hazards. "#0475". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Oxygen difluoride". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Lebeau, P.; Damiens, A. (1929). "Sur un nouveau mode de préparation du fluorure d'oxygène" [A new method of preparation of oxygen fluoride]. Comptes rendus hebdomadaires des séances de l'Académie des Sciences (in French). 188: 1253–1255. Retrieved February 21, 2013.
- ^ Lebeau, P.; Damiens, A. (1927). "Sur l'existence d'un composé oxygéné du fluor" [The existence of an oxygen compound of fluorine]. Comptes rendus hebdomadaires des séances de l'Académie des Sciences (in French). 185: 652–654. Retrieved February 21, 2013.
External links[]
- Oxygen compounds
- Fluorides
- Nonmetal halides
- Rocket oxidizers
- Oxidizing agents
- Chalcohalides