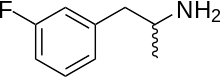
3-Fluoroamphetamine
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C9H12FN |
| Molar mass | 153.200 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
3-Fluoroamphetamine (3-FA; PAL-353) is a stimulant drug from the amphetamine family which acts as a monoamine releaser with similar potency to methamphetamine but more selectivity for dopamine and norepinephrine release over serotonin.[1] It is self-administered by mice to a similar extent to related drugs such as 4-fluoroamphetamine and 3-methylamphetamine.[2]
Legal status[]
China[]
As of October 2015 3-FA is a controlled substance in China.[3]
See also[]
- 2-Fluoroamphetamine (2-FA)
- 3-Fluoroethamphetamine (3-FEA)
- 3-Fluoromethamphetamine (3-FMA)
- 3-Hydroxyamphetamine (Gepefrine)
- 3-Methylamphetamine (3-MA)
- 3-Methoxyamphetamine (3-MeOA)
- 3-Trifluoromethylamphetamine (Norfenfluramine)
- 4-Fluoroamphetamine (4-FA)
References[]
- ^ Negus SS, Mello NK, Blough BE, Baumann MH, Rothman RB (February 2007). "Monoamine releasers with varying selectivity for dopamine/norepinephrine versus serotonin release as candidate "agonist" medications for cocaine dependence: studies in assays of cocaine discrimination and cocaine self-administration in rhesus monkeys". The Journal of Pharmacology and Experimental Therapeutics. 320 (2): 627–36. doi:10.1124/jpet.106.107383. PMID 17071819. S2CID 8326027.
- ^ Wee S, Anderson KG, Baumann MH, Rothman RB, Blough BE, Woolverton WL (May 2005). "Relationship between the serotonergic activity and reinforcing effects of a series of amphetamine analogs". The Journal of Pharmacology and Experimental Therapeutics. 313 (2): 848–54. doi:10.1124/jpet.104.080101. PMID 15677348. S2CID 12135483.
- ^ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Archived from the original on 1 October 2015. Retrieved 1 October 2015.
Categories:
- Substituted amphetamines
- Fluoroarenes
- Designer drugs
- Norepinephrine-dopamine releasing agents