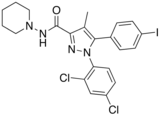
AM-251
1-(2,4-Dichlorophenyl)-5-(4-iodophenyl)-4-methyl-N -(1-piperidyl)pyrazole-3-carboxamide
CAS Number PubChem CID IUPHAR/BPS ChemSpider UNII ChEBI ChEMBL CompTox Dashboard (EPA ) ECHA InfoCard 100.162.062 Formula C 22 H 21 Cl 2 I N 4 O Molar mass −1 3D model (JSmol )
O=C(NN1CCCCC1)c4nn(c2ccc(Cl)cc2Cl)c(c3ccc(I)cc3)c4C
InChI=1S/C22H21Cl2IN4O/c1-14-20(22(30)27-28-11-3-2-4-12-28)26-29(19-10-7-16(23)13-18(19)24)21(14)15-5-8-17(25)9-6-15/h5-10,13H,2-4,11-12H2,1H3,(H,27,30)
Y Key:BUZAJRPLUGXRAB-UHFFFAOYSA-N
Y
AM-251 is an inverse agonist at the CB1 cannabinoid receptor. AM-251 is structurally very close to rimonabant ; both are cannabinoid receptor antagonists . In AM-251, the p -chloro group attached to the phenyl substituent at C-5 of the pyrazole ring is replaced with a p -iodo group. The resulting compound exhibits slightly better binding affinity for the CB1 receptor (with a Ki value of 7.5 nM) than rimonabant, which has a Ki value of 11.5 nM, AM-251 is, however, about two-fold more selective for the CB1 receptor when compared to rimonabant.[1] [2] [3]
AM251 has shown an in vitro pancreatic and colon cancer cells.[4]
See also [ ] Discovery and development of Cannabinoid Receptor 1 Antagonists References [ ]
^ Lan R, Liu Q, Fan P, Lin S, Fernando SR, McCallion D, et al. (February 1999). "Structure-activity relationships of pyrazole derivatives as cannabinoid receptor antagonists". Journal of Medicinal Chemistry . 42 (4): 769–76. doi :10.1021/jm980363y . PMID 10052983 . ^ Seely KA, Brents LK, Franks LN, Rajasekaran M, Zimmerman SM, Fantegrossi WE, Prather PL (October 2012). "AM-251 and rimonabant act as direct antagonists at mu-opioid receptors: implications for opioid/cannabinoid interaction studies" . Neuropharmacology . 63 (5): 905–15. doi :10.1016/j.neuropharm.2012.06.046 . PMC 3408547 PMID 22771770 . ^ Seely, Kathryn A.; Brents, Lisa K.; Franks, Lirit N.; Rajasekaran, Maheswari; Zimmerman, Sarah M.; Fantegrossi, William E.; Prather, Paul L. (October 2012). "AM-251 and rimonabant act as direct antagonists at mu-opioid receptors: Implications for opioid/cannabinoid interaction studies" . Neuropharmacology . 63 (5): 905–915. doi :10.1016/j.neuropharm.2012.06.046 . PMC 3408547 PMID 22771770 . ^ Carpi, Sara; Fogli, Stefano; Romanini, Antonella; Pellegrino, Mario; Adinolfi, Barbara; Podestà, Adriano; Costa, Barbara; Da Pozzo, Eleonora; Martini, Claudia; Breschi, Maria Cristina; Nieri, Paola (August 26, 2015). "AM251 induces apoptosis and G2/M cell cycle arrest in A375 human melanoma cells" (PDF) . Anticancer Drugs . 26 (7): 754–62. doi :10.1097/CAD.0000000000000246 . PMID 25974027 . S2CID 205526223 . Archived (PDF) from the original on July 2, 2021. Retrieved July 3, 2021 .
Cannabinoids
Phytocannabinoids
Cannabichromenes Cannabicyclols Cannabidiols Cannabielsoins Cannabigerols Cannabinols and cannabinodiols Cannabitriols Delta-8-tetrahydrocannabinols Delta-9-tetrahydrocannabinols
Delta-9-THC (THC)
THCH THCP
THCV
Miscellaneous cannabinoids Active metabolites
Endocannabinoids
Arachidonoyl ethanolamide (AEA; anandamide) 2-Arachidonoylglycerol (2-AG) 2-Arachidonyl glyceryl ether (2-AGE; noladin ether) 2-Oleoylglycerol (2-OG) N-Arachidonoyl dopamine (NADA) N-Arachidonylglycine (NAGly) N-Arachidonoyl serotonin (AA-5-HT) Docosatetraenoylethanolamide (DEA) Lysophosphatidylinositol (LPI) Oleamide Oleoylethanolamide (OEA) Palmitoylethanolamide (PEA) RVD-Hpα Stearoylethanolamide (SEA) O-Arachidonoyl ethanolamine (O-AEA; virodhamine) Synthetic
Classical cannabinoids Non-classical Adamantoylindoles Benzimidazoles Benzoylindoles
AM-630 AM-679 AM-694 AM-1241 AM-2233 GW-405,833 (L-768,242) Pravadoline RCS-4 WIN 54,461 Cyclohexylphenols Eicosanoids
ACEA ACPA Methanandamide (AM-356) O-1812 Hydrocarbons Indazole carboxamides Indazole-3- Indole-3-carboxamides Indole-3-carboxylates Naphthoylindazoles Naphthoylindoles Naphthoylpyrroles Naphthylmethylindenes Naphthylmethylindoles Phenylacetylindoles Pyrazolecarboxamides Pyrrolobenzoxazines Quinolinyl esters Tetramethylcyclo- Tetramethylcyclo-
A-796,260 A-834,735 FUB-144 UR-144 XLR-11 XLR-12 Tetramethylcyclo- Others
Allosteric CBR ligands Endocannabinoid (inactivation inhibitors) Anticannabinoids (antagonists/inverse
AM-251 AM-630 AM-6545 BML-190 Drinabant (AVE1625) Hemopressin Ibipinabant (SLV319) JTE-907 LY-320,135 MK-9470 NESS-0327 O-1918 O-2050 Otenabant (CP-945,598) PF-514273 PipISB PSB-SB-487 Rimonabant (SR141716) Rosonabant (E-6776) SR-144,528 Surinabant (SR147778) Taranabant (MK-0364) TM-38837 VCHSR
See also: Cannabinoid receptor modulators (cannabinoids by pharmacology)List of: AM cannabinoids JWH cannabinoids Designer drugs § Synthetic cannabimimetics
Receptor (ligands )
CB1
Agonists (abridged; see here for more) : 2-AG 2-AGE (noladin ether) 11-Hydroxy-THC α-Amyrin · β-Amyrin AB-CHMINACA AM-1220 AM-1221 AM-1235 AM-2201 AM-2232 Anandamide AZ-11713908 Cannabinol CB-13 CP 47,497 CP 55,940 Dimethylheptylpyran DEA ECG EGCG Epicatechin Gallocatechol (gallocatechin) Honokiol HU-210 JWH-007 JWH-015 JWH-018 JWH-073 Kavain L-759,633 Levonantradol Menabitan Nabilone Nabitan NADA O-1812 Oleamide Pravadoline Serinolamide A THC (dronabinol) UR-144 WIN 55,212-2 Yangonin Antagonists: AM-251 AM-6545 Cannabidiol Cannabigerol Drinabant Falcarinol (carotatoxin) Hemopressin Ibipinabant LY-320,135 MK-9470 NESS-0327 O-2050 Otenabant PF-514273 PipISB Rimonabant Rosonabant Surinabant Taranabant THCV TM-38837 VCHSR Virodhamine CB2
Agonists: 2-AG 2-AGE (noladin ether) 3,3'-Diindolylmethane 4-O-Methylhonokiol α-Amyrin · β-Amyrin A-796,260 A-834,735 A-836,339 AM-1221 AM-1235 AM-1241 AM-2232 Anandamide AZ-11713908 Cannabinol Caryophyllene CB-13 CBS-0550 CP 55,940 GW-405,833 (L-768,242) GW-842,166X HU-308 JTE 7-31 JWH-007 JWH-015 JWH-018 JWH-133 L-759,633 L-759,656 Magnolol MDA-19 Nabitan NADA Olorinab (APD-371) PF-03550096 S-444,823 SER-601 Serinolamide A UR-144 Tedalinab THC (dronabinol) THCV Virodhamine Antagonists: 4-O-Methylhonokiol AM-630 BML-190 Cannabidiol Honokiol JTE-907 SR-144,528 WIN 54,461 WIN 56,098 NAGly GPR18 )
Agonists: Abnormal cannabidiol ACPA AM251 Anandamide Cannabidiol NADGly THC (dronabinol) O-1602 GPR55
Agonists: 2-AGE (noladin ether) Abnormal cannabidiol AM-251 CP 55,940 Lysophosphatidylinositol O-1602 Oleoylethanolamide Palmitoylethanolamide THC (dronabinol) GPR119 Unsorted
Transporter (modulators )
eCBTs
Inhibitors: AM-404 Arachidonoyl serotonin Cannabidiol Guineensine LY-2183240 Paracetamol (acetaminophen) URB-597 VDM-11
Enzyme (modulators )
FAAH MAGL
Inhibitors: IDFP JZL-184 JZL-195 MAFP URB-602 ABHD6 ABHD12
Inhibitors: Betulinic acid Maslinic acid MAFP Oleanolic acid Orlistat (tetrahydrolipstatin) Ursolic acid
Others
Precursors: Phosphatidylethanolamine NAPE Diacylglycerol Others: (directly potentiates activity of 2-AG at CB1 receptor) (FAAH-like anandamide transporter inhibitor)
See also
Receptor/signaling modulators Cannabinoids (cannabinoids by structure)
MOR DOR KOR NOP
Agonists: Buprenorphine Cebranopadol Dihydroetorphine Etorphine Levomethorphan Levorphanol MCOPPB NNC 63-0532 Nociceptin (orphanin FQ) Norbuprenorphine Racemethorphan Racemorphan Ro64-6198 SR-16435 Antagonists: AT-076 BTRX-246040 (LY-2940094) J-113,397 JTC-801 SB-612,111 Thienorphine Unsorted
β-Casomorphins Amidorphin Cytochrophin-4 Gliadorphin (gluteomorphin) Gluten exorphins Hemorphins Kava constituentsNEM Neoendorphins Nepetalactone (catnip )Rubiscolins Others
Enkephalinase inhibitors :Amastatin Candoxatril D -PhenylalanineEcadotril (sinorphan) Kelatorphan Racecadotril (acetorphan) RB-101 RB-120 RB-3007 Selank Semax Spinorphin Thiorphan Tynorphin Ubenimex (bestatin) Propeptides: β-Lipotropin (proendorphin) Prodynorphin Proenkephalin Proopiomelanocortin (POMC) Others: Kyotorphin (met-enkephalin releaser/degradation stabilizer)