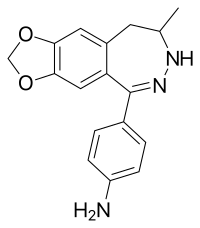
GYKI 52895 Other names 4-(8,9-Dihydro-8-methyl-7H -1,3-dioxolo[4,5-h ][2,3]benzodiazepin-5-yl)benzenamine
4-{13-methyl-4,6-dioxa-11,12-diazatricyclo[7.5.0.03,7 ]tetradeca-1,3(7),8,10-tetraen-10-yl}aniline
CAS Number PubChem CID ChemSpider UNII KEGG ChEMBL CompTox Dashboard (EPA ) Formula C 17 H 17 N 3 O 2 Molar mass −1 3D model (JSmol )
O1c2c(OC1)cc3c(c2)C(=N/NC(C3)C)\c4ccc(N)cc4
InChI=1S/C17H17N3O2/c1-10-6-12-7-15-16(22-9-21-15)8-14(12)17(20-19-10)11-2-4-13(18)5-3-11/h2-5,7-8,10,19H,6,9,18H2,1H3
Y Key:AQTITSBNGSVQNZ-UHFFFAOYSA-N
Y
GYKI 52895 is a drug which is a 2,3-benzodiazepine derivative that also shares the 3,4-methylenedioxyphenethylamine pharmacophore . Unlike other similar drugs, GYKI 52895 is a selective dopamine reuptake inhibitor (DARI),[1] [2] [3] amphetamine and its derivatives (e.g. dextromethamphetamine ), cocaine , and methylphenidate and its derivatives (e.g. ethylphenidate ). However, dopaminergic drugs are also prone to producing emetic effects such as in the case of apomorphine .
Egis Pharmaceuticals began clinical development of the drug in 1997 for major depressive disorder and Parkinson's disease , but it was discontinued in 2001.[4]
See also [ ] GYKI 52466 , another 2,3-benzodiazepine with other than GABAergic functionTifluadom Lufuradom Benzodiazepine Substituted methylenedioxyphenethylamine References [ ]
^ Horváth K, Szabó H, Pátfalusi M, Berzsenyi P, Andrási F (1990). "Pharmacological Effects of GYKI 52895, a New Selective Dopamine Uptake Inhibitor". European Journal of Pharmacology . 183 (4): 1416–1417. ^ Huang CL, Chen HC, Huang NK, Yang DM, Kao LS, Chen JC, et al. (June 1999). "Modulation of dopamine transporter activity by nicotinic acetylcholine receptors and membrane depolarization in rat pheochromocytoma PC12 cells" . Journal of Neurochemistry . 72 (6): 2437–44. doi :10.1046/j.1471-4159.1999.0722437.x PMID 10349853 . ^ Vaarmann A, Gandhi S, Gourine AV, Abramov AY (2010). "Novel pathway for an old neurotransmitter: dopamine-induced neuronal calcium signalling via receptor-independent mechanisms". Cell Calcium . 48 (2–3): 176–82. doi :10.1016/j.ceca.2010.08.008 . PMID 20846720 . ^ "GYKI 52895" . Adis Insight.
Stimulants
Adamantanes
Adapromine Amantadine Bromantane Memantine Rimantadine Adenosine antagonists Alkylamines
Cyclopentamine Cypenamine Cyprodenate Heptaminol Isometheptene Levopropylhexedrine Methylhexaneamine Octodrine Propylhexedrine Tuaminoheptane Ampakines Arylcyclohexylamines Benzazepines
6-Br-APB SKF-77434 SKF-81297 SKF-82958 Cathinones Cholinergics Convulsants Eugeroics Oxazolines Phenethylamines Phenylmorpholines Piperazines
2C-B-BZP 3C-PEP BZP CM156 DBL-583 GBR-12783 GBR-12935 GBR-13069 GBR-13098 GBR-13119 MeOPP MBZP oMPP Vanoxerine Piperidines
1-Benzyl-4-(2-(diphenylmethoxy)ethyl)piperidine 2-Benzylpiperidine 2-Methyl-3-phenylpiperidine 3,4-Dichloromethylphenidate 4-Benzylpiperidine 4-Fluoromethylphenidate 4-Methylmethylphenidate Desoxypipradrol Difemetorex Diphenylpyraline Ethylnaphthidate Ethylphenidate Methylnaphthidate Isopropylphenidate JZ-IV-10 Methylphenidate (Dexmethylphenidate )Nocaine Phacetoperane Pipradrol Propylphenidate SCH-5472 Pyrrolidines Racetams
Oxiracetam Phenylpiracetam Phenylpiracetam hydrazide Tropanes Tryptamines Others ATC code : N06B
Monoamine reuptake inhibitors
DAT (DRIs
Piperazines: DBL-583 GBR-12783 GBR-12935 GBR-13069 GBR-13098 Nefazodone Vanoxerine
Piperidines: 4-Fluoropethidine Benocyclidine (BTCP) Desoxypipradrol Dexmethylphenidate Difemetorex Ethylphenidate HDMP-28 Methylphenidate Pethidine (meperidine) Phencyclidine Pipradrol Serdexmethylphenidate Tenocyclidine
Pyrrolidines: Diphenylprolinol MDPV Naphyrone Prolintane Pyrovalerone
Tropanes: Altropane Benzatropine (benztropine) Brasofensine CFT Cocaine Dichloropane Difluoropine Etybenzatropine (ethybenztropine) FE-β-CPPIT FP-β-CPPIT Ioflupane (123 I) RTI-55 RTI-112 RTI-113 RTI-121 RTI-126 RTI-150 RTI-177 RTI-229 RTI-336 Tesofensine Troparil Tropoxane WF-11 WF-23 WF-31 WF-33
NET (NRIs
Norepinephrine–dopamine reuptake inhibitors: Amineptine Bupropion Fencamine Fencamfamin Hydroxybupropion Lefetamine Levophacetoperane LR-5182 Manifaxine Methylphenidate Nomifensine O-2172 Radafaxine Serdexmethylphenidate Solriamfetol
Tricyclic antidepressants: Amitriptyline Butriptyline Cianopramine Clomipramine Desipramine Dosulepin (dothiepin) Doxepin Imipramine Lofepramine Melitracen Nortriptyline Protriptyline Trimipramine
Tetracyclic antidepressants: Amoxapine Maprotiline Mianserin Oxaprotiline Setiptiline
Others: Antihistamines (e.g., brompheniramine , chlorphenamine , pheniramine , tripelennamine )Antipsychotics (e.g., loxapine , ziprasidone )Arylcyclohexylamines (e.g., ketamine , phencyclidine )Dopexamine Ephenidine Ginkgo biloba Indeloxazine Nefazodone Opioids (e.g., desmetramadol , methadone , pethidine (meperidine) , tapentadol , tramadol , levorphanol )
SERT (SRIs
Selective serotonin reuptake inhibitors and serotonin receptor modulators: Etoperidone Litoxetine Lubazodone LY-393558 Quipazine SB-649915 TGBA01AD Trazodone Vilazodone Vortioxetine
Serotonin–norepinephrine reuptake inhibitors: Atomoxetine (tomoxetine) Bicifadine Desvenlafaxine Duloxetine Eclanamine Levomilnacipran McN5652 Milnacipran N-Methyl-PPPA PPPA Tofenacin Venlafaxine
Tricyclic antidepressants: Amitriptyline Cianopramine Clomipramine Cyanodothiepin Desipramine Dosulepin (dothiepin) Doxepin Imipramine Lofepramine Nortriptyline Pipofezine Protriptyline
Others: Amoxapine Antihistamines (e.g., brompheniramine , chlorphenamine , dimenhydrinate , diphenhydramine , mepyramine (pyrilamine) , pheniramine , tripelennamine )Antipsychotics (e.g., loxapine , ziprasidone )Arylcyclohexylamines (e.g., 3-MeO-PCP , esketamine , ketamine , methoxetamine , phencyclidine )Cyclobenzaprine Delucemine Dextromethorphan Dextrorphan Efavirenz Hypidone Medifoxamine Mesembrine Mifepristone MIN-117 (WF-516) N-Me-5-HT Opioids (e.g., dextropropoxyphene , methadone , pethidine (meperidine) , levorphanol , tapentadol , tramadol )Roxindole
VMATs
Amiodarone Amphetamines (e.g., amphetamine , methamphetamine , MDMA )Bietaserpine Deserpidine Deutetrabenazine Dihydrotetrabenazine Efavirenz GBR-12935 Ibogaine Ketanserin Lobeline Reserpine Rose bengal Tetrabenazine Valbenazine Vanoxerine (GBR-12909) Others
DAT modulators: Agonist-like: SoRI-9804 ; Antagonist-like: SoRI-20041 See also: Receptor/signaling modulators • Monoamine releasing agents • Adrenergics • Dopaminergics • Serotonergics • Monoamine metabolism modulators • Monoamine neurotoxins