Ubenimex[1]
Names
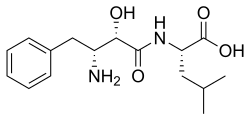
Preferred IUPAC name
(2S )-2-[(2S ,3R )-3-Amino-2-hydroxy-4-phenylbutanamido]-4-methylpentanoic acid
Other names
Bestatin; N -[(2S ,3R )-3-Amino-2-hydroxy-4-phenylbutyryl]-L -leucine
Identifiers
CAS Number
3D model (JSmol )
ChEMBL
ChemSpider
ECHA InfoCard 100.055.917
UNII
InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13+,14+/m1/s1
Key: VGGGPCQERPFHOB-RDBSUJKOSA-N
CC(C)C[C@@H](C(=O)O)NC(=O)[C@H]([C@@H](CC1=CC=CC=C1)N)O
Properties
Chemical formula
C 16 H 24 N 2 O 4
Molar mass
−1
Melting point
245 °C (473 °F; 518 K) (decomposes)
Hazards
S-phrases (outdated) S22 S24/25
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Y what is Y N
Infobox references
Chemical compound
Ubenimex (INN ), also known more commonly as bestatin , is a competitive, reversible protease inhibitor . It is an inhibitor of arginyl aminopeptidase (aminopeptidase B),[2] leukotriene A4 hydrolase (a zinc metalloprotease that displays both epoxide hydrolase and aminopeptidase activities),[3] alanyl aminopeptidase (aminopeptidase M/N),[4] leucyl/cystinyl aminopeptidase (oxytocinase/vasopressinase),[5] [6] membrane dipeptidase (leukotriene D4 hydrolase). It is being studied for use in the treatment of acute myelocytic leukemia [7] lymphedema .[8] Streptomyces olivoreticuli [9] oxytocin , vasopressin , enkephalins , and various other peptides and compounds.[citation needed
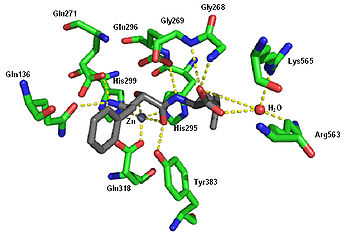
Crystal structure of ubenimex in the binding pocket of leukotriene A4 hydrolase. Rendered from PDB
1HS6 .
See also [ ] References [ ]
^ N-((2S,3R)-3-Amino-2-hydroxy-4-phenylbutyryl)-L-leucine at Sigma-Aldrich ^ Umezawa, H.; Aoyagi, T.; Suda, H.; Hamada, M.; Takeuchi, T. (1976). "Bestatin, an inhibitor of aminopeptidase B, produced by actinomycetes" . The Journal of Antibiotics . 29 (29): 97–99. doi :10.7164/antibiotics.29.97 PMID 931798 . ^ Muskardin, D.T.; Voelkel, N.F.; Fitzpatrick, F.A. (1994). "Modulation of pulmonary leukotriene formation and perfusion pressure by Bestatin, an inhibitor of leukotriene A4 hydrolase". Biochemical Pharmacology . 48 (48): 131–137. doi :10.1016/0006-2952(94)90232-1 . PMID 8043014 . ^ K Sekine; H Fujii; F Abe (1999). "Induction of apoptosis by Bestatin (ubenimex) in human leukemic cell lines" . Leukemia . 13 (5): 729–734. doi :10.1038/sj.leu.2401388 PMID 10374877 . ^ Nakanishi Y, Nomura S, Okada M, Ito T, Katsumata Y, Kikkawa F, Hattori A, Tsujimoto M, Mizutani S (2000). "Immunoaffinity purification and characterization of native placental leucine aminopeptidase/oxytocinase from human placenta". Placenta . 21 (7): 628–34. doi :10.1053/plac.2000.0564 . PMID 10985965 . ^ Naruki M, Mizutani S, Goto K, Tsujimoto M, Nakazato H, Itakura A, Mizuno K, Kurauchi O, Kikkawa F, Tomoda Y (1996). "Oxytocin is hydrolyzed by an enzyme in human placenta that is identical to the oxytocinase of pregnancy serum". Peptides . 17 (2): 257–61. doi :10.1016/0196-9781(95)02124-8 . PMID 8801531 . S2CID 28486489 . ^ Hirayama, Y; Sakamaki, S; Takayanagi, N; Tsuji, Y; Sagawa, T; Chiba, H; Matsunaga, T; Niitsu, Y (2003). "Chemotherapy with ubenimex corresponding to patient age and organ disorder for 18 cases of acute myelogeneous leukemia in elderly patients--effects, complications and long-term survival". Gan to Kagaku Ryoho. Cancer & Chemotherapy . 30 (8): 1113–8. PMID 12938265 . ^ Tian, W; Rockson, S; Jiang, X; Kim, J; Begaye, A; Shuffle, EM; Tu, AB; Cribb, M; Nepiyushchikh, Z; Feroze, AH; Zamanian, RT; Dhillon, RT; Voelkel, NF; Peters-Golden, M; Kitajewski, J; Dixon, JB; Nicolls, MR (2017). "Leukotriene B4 antagonism ameliorates experimental lymphedema" . Science Translational Medicine . 9 (389): eaal3920. doi :10.1126/scitranslmed.aal3920 PMID 28490670 . ^ Bauvois, B; Dauzonne, D (January 2006). "Aminopeptidase-N/CD13 (EC 3.4.11.2) inhibitors: Chemistry, biological evaluations, and therapeutic prospects" . Medicinal Research Reviews . 26 (1): 88–130. doi :10.1002/med.20044 . PMC 7168514 PMID 16216010 .
External links [ ] The MEROPS online database for peptidases and their inhibitors: Bestatin
Receptor (ligands )
BLT
BLT1
Agonists: 12-HETE Leukotriene B4 BLT2
Agonists: 12-HETE 12-HHT 15-HETE Leukotriene B4
CysLT
CysLT1
Agonists: Leukotriene C4 Leukotriene D4 Leukotriene E4 Antagonists: Ablukast Montelukast Pranlukast Zafirlukast CysLT2
Agonists: Leukotriene C4 Leukotriene D4 Leukotriene E4 CysLTE
Enzyme (inhibitors )
Others
Precursors: Linoleic acid γ-Linolenic acid (gamolenic acid) Dihomo-γ-linolenic acid Diacylglycerol Arachidonic acid 5-HPETE (arachidonic acid 5-hydroperoxide) Leukotriene A4
See also
Receptor/signaling modulators Prostanoid signaling modulators
Adiponectin
AdipoR1 AdipoR2
Agonists: Peptide: Adiponectin ; Non-peptide: AdipoRon
Deoxyschizandrin Parthenolide Taxifoliol
Angiotensin Bradykinin
Agonists: Bradykinin Kallidin Antagonists: Icatibant LF22-0542 CGRP
Agonists: Amylin CGRP Pramlintide Antibodies: Eptinezumab Erenumab Fremanezumab Galcanezumab Cholecystokinin
CCKA
Agonists: Cholecystokinin CCKB
Agonists: Cholecystokinin CCK-4 Gastrin Pentagastrin (CCK-5) Unsorted
CRH
CRF1
Agonists: Corticorelin Corticotropin-releasing hormone Sauvagine Urocortin CRF2
Agonists: Corticorelin Corticotropin-releasing hormone Sauvagine Urocortin
Cytokine See here instead.
Endothelin
Agonists: Endothelin 1 Endothelin 2 Endothelin 3 Sarafotoxin Galanin
GAL1
Agonists: Galanin Galanin-like peptide Galmic Galnon GAL2
Agonists: Galanin Galanin-like peptide Galmic Galnon GAL3
Agonists: Galanin Galmic Galnon
Ghrelin/GHS GH GHRH GLP
GLP-1 GLP-2
Agonists: GLP-2 Teduglutide Others
Glucagon GnRH Gonadotropin Growth factor See here instead.
Insulin
Agonists: Chaetochromin (4548-G05) Insulin-like growth factor 1 Insulin-like growth factor 2 Insulin Insulin aspart Insulin degludec Insulin detemir Insulin glargine Insulin glulisine Insulin lispro Mecasermin Mecasermin rinfabate Kinase inhibitors: Linsitinib Antibodies: (against IGF-1 and IGF-2) Kisspeptin Leptin
Agonists: Leptin Metreleptin MCH
MCH1
Agonists: Melanin-concentrating hormone MCH2
Agonists: Melanin-concentrating hormone
Melanocortin Neuropeptide FF
Agonists: Neuropeptide FF Neuropeptide S Neuropeptide Y
Y1
Agonists: Neuropeptide Y Peptide YY Y2
Agonists: Neuropeptide Y Peptide YY Y4
Agonists: Neuropeptide Y Pancreatic polypeptide Peptide YY Y5
Agonists: Neuropeptide Y Peptide YY
Neurotensin
NTS1
Agonists: Neurotensin Neuromedin N NTS2
Opioid See here instead.
Orexin Oxytocin Prolactin PTH
Agonists: Abaloparatide Parathyroid hormone Parathyroid hormone-related protein (PTHrP) Teriparatide Relaxin
Agonists: Insulin-like factor 3 Relaxin (, , 3 )Serelaxin Somatostatin Tachykinin TRH TSH
Agonists: Thyrotropin alfa TSH (thyrotropin) Vasopressin VIP /PACAP
VIPR1
Agonists: Peptide: PACAP VIP VIPR2 PAC1 Unsorted
Others
Endogenous: Adrenomedullin Apelin Asprosin Bombesin Calcitonin Carnosine CART CLIP DSIP Enteroglucagon Formyl peptide GALP GIP GRP Integrin ligands (collagens , fibrinogen , fibronectin , laminins , ICAM-1 , ICAM-2 , osteopontin , VCAM-1 , vitronectin )Kininogens Motilin Natriuretic peptides (ANP , BNP , CNP , urodilatin )Nesfatin-1 Neuromedin B Neuromedin N Neuromedin S Neuromedin U Obestatin Osteocalcin Resistin Secretin Thymopoietin Thymosins Thymulin Urotensin-II VGF Exogenous: Lifitegrast (LFA-1 antagonist)
See also
Receptor/signaling modulators
MOR
Unknown/unsorted: Cannabidiol Coronaridine Cyproterone acetate Tabernanthine Tetrahydrocannabinol DOR
Unknown/unsorted: 18-MC Cannabidiol Coronaridine Cyproterone acetate Tabernanthine Tetrahydrocannabinol KOR
Unknown/unsorted: Akuammicine Akuammine Coronaridine Cyproterone acetate Ibogamine Tabernanthine NOP Unsorted
β-Casomorphins Amidorphin Cytochrophin-4 Gliadorphin (gluteomorphin) Gluten exorphins Hemorphins Kava constituentsNEM Neoendorphins Nepetalactone (catnip )Rubiscolins Others
Propeptides: β-Lipotropin (proendorphin) Prodynorphin Proenkephalin Proopiomelanocortin (POMC) Others: Kyotorphin (met-enkephalin releaser/degradation stabilizer)