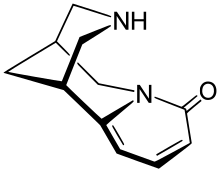
Cytisine
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
(1R,5S)-1,2,3,4,5,6-Hexahydro-8H-1,5-methanopyrido[1,2-a][1,3]diazocin-8-one | |
| Other names
Cytisine
Baptitoxine Sophorine | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.924 |
IUPHAR/BPS
|
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
show
InChI | |
show
SMILES | |
| Properties | |
Chemical formula
|
C11H14N2O |
| Molar mass | 190.24 g/mol |
| Melting point | 152 to 153 °C (306 to 307 °F; 425 to 426 K) |
| Boiling point | 218 °C (424 °F; 491 K) at 2 mmHg |
| Pharmacology | |
ATC code
|
N07BA04 (WHO) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cytisine, also known as baptitoxine and sophorine, is an alkaloid that occurs naturally in several plant genera, such as Laburnum and Cytisus of the family Fabaceae. It has been used medically to help with smoking cessation.[1] Although widely used for smoking cessation in Eastern Europe, cytisine remains relatively unknown beyond it. However, it has been found effective in several randomized clinical trials, including some in the United States and a large one in New Zealand,[1] and is currently being investigated in additional trials in the United States (being conducted by Achieve Life Sciences) and a non-inferiority trial in Australia in which it is being compared head-to-head with the smoking cessation aid varenicline (sold in the United States as Chantix).[2]
Sources[]
Cytisine is extracted from the seeds of L. (Golden Rain acacia), and is found in several genera of the subfamily Faboideae of the family Fabaceae, including Laburnum, Anagyris, Thermopsis, Cytisus, Genista, Retama and Sophora. Cytisine is also present in Gymnocladus of the subfamily Caesalpinioideae.
Uses[]
Smoking cessation[]
Cytisine has been available in post-Soviet states for more than 40 years as an aid to smoking cessation under the brand name Tabex from the Bulgarian pharmaceutical company Sopharma AD. It was first marketed in Bulgaria in 1964 and then became widely available the then-Soviet Union.[citation needed] In Poland, it is sold under the brand name Desmoxan, and it is also available in Canada under the brand name Cravv.
Its molecular structure has some similarity to that of nicotine, and it has similar pharmacological effects. Like the smoking cessation aid varenicline, cytisine is a partial agonist of nicotinic acetylcholine receptors (nAChRs).[3] Cytisine has a short half-life of 4.8 hours,[4] As a result, the extract provides smokers with satisfaction similar to smoking a cigarette, alleviating the urge to smoke and reducing the severity of nicotine withdrawal symptoms, while also reducing the reward experience of any cigarettes smoked.[5]
Cytisine is rapidly eliminated from the body.
In 2011, a randomized controlled trial with 740 patients found cytisine improved 12-month abstinence from nicotine from 2.4% with placebo to 8.4% with cytisine.[6] A 2013 meta-analysis of eight studies demonstrated that cytisine has similar effectiveness to varenicline but with substantially lower side effects.[7] A 2014 systematic review and economic evaluation concluded that cytisine was more likely to be cost-effective for smoking cessation than varenicline.[8]
Recreational[]
Plants containing cytisine, including the scotch broom and mescalbean, have also been used recreationally. Positive effects are reported to include a nicotine-like intoxication.[6]
Reagent for organic chemistry[]
(-)-Cytisine extracted from Laburnum anagyroides seeds was used as a starting material for the preparation of "(+)-sparteine surrogate," for the preparation of enantiomerically enriched lithium anions of opposite stereochemistry to those anions obtained from sparteine.[9]
Toxicity[]
Cytisine can interfere with breathing and cause death; LD50 i.v., in mice is about 2 mg/kg.[10] Cytisine is also teratogenic.[11]
Māmane (Sophora chrysophylla) can contain amounts of cytisine that are lethal to most animals. The palila (Loxioides bailleui, a bird), Uresiphita polygonalis virescens and Cydia species (moths), and possibly sheep and goats are not affected by the toxin for various reasons, and use māmane, or parts thereof, as food. U. p. virescens caterpillars are possibly able to sequester the cytisine to give themselves protection from getting eaten; they have aposematic coloration which would warn off potential predators.[12]
References[]
- ^ Jump up to: a b Walker N, Howe C, Glover M, McRobbie H, Barnes J, Nosa V, Parag V, Bassett B, Bullen C (2014). "Cytisine versus Nicotine for Smoking Cessation". New England Journal of Medicine. 371 (25): 2353–62. doi:10.1056/nejmoa1407764. PMID 25517706.
- ^ Thomas D, Farrell M, McRobbie H, Tutka P, Petrie D, West R, Siahpush M, Gartner C, Walker N, Mendelsohn CP, Hall W, Paul C, Zwar N, Ferguson SG, Boland VC, Richmond R, Doran CM, Shakeshaft A, Mattick RP, Courtney RJ (May 2019). "The effectiveness, safety and cost-effectiveness of cytisine versus varenicline for smoking cessation in an Australian population: a study protocol for a randomized controlled non-inferiority trial". Addiction. 114 (5): 923–933. doi:10.1111/add.14541. Retrieved 4 June 2021.CS1 maint: multiple names: authors list (link)
- ^ Dallanoce, Clelia; Frigerio, Fabio; Martelli, Giuliana; Grazioso, Giovanni; Matera, Carlo; Pomè, Diego Yuri; Pucci, Luca; Clementi, Francesco; Gotti, Cecilia; Amici, Marco De (2010). "Novel tricyclic Δ2-isoxazoline and 3-oxo-2-methyl-isoxazolidine derivatives: Synthesis and binding affinity at neuronal nicotinic acetylcholine receptor subtypes". Bioorganic & Medicinal Chemistry. 18 (12): 4498–4508. doi:10.1016/j.bmc.2010.04.065. ISSN 0968-0896. PMID 20478710.
- ^ Jeong, Soo Hee; Newcombe, David; Sheridan, Janie; Tingle, Malcolm (2015-06-01). "Pharmacokinetics of cytisine, an α4β2 nicotinic receptor partial agonist, in healthy smokers following a single dose". Drug Testing and Analysis. 7 (6): 475–482. doi:10.1002/dta.1707. ISSN 1942-7611. PMID 25231024.
- ^ Reinberg, Steven (December 17, 2014). "Cheap Natural Compound May Help Smokers Quit. But cytisine isn't widely available, study authors note". WebMD. Retrieved 4 June 2021.
- ^ Jump up to: a b West R, Zatonski W, Cedzynska M, Lewandowska D, Pazik J, Aveyard P, Stapleton J (2011). "Placebo-Controlled Trial of Cytisine for Smoking Cessation". New England Journal of Medicine. 365 (13): 1193–1200. doi:10.1056/NEJMoa1102035. PMID 21991893.
- ^ Hajek, McRobbie and Myers (25 Feb 2013). "Efficacy of cytisine in helping smokers quit: systematic review and meta-analysis". Thorax. 68 (11): 1037–42. doi:10.1136/thoraxjnl-2012-203035. PMID 23404838.
- ^ Leaviss, Joanna; Sullivan, William; Ren, Shijie; Everson-Hock, Emma; Stevenson, Matt; Stevens, John; Strong, Mark; Cantrell, Anna (2014). "What is the clinical effectiveness and cost-effectiveness of cytisine compared with varenicline for smoking cessation? A systematic review and economic evaluation". Health Technology Assessment. 18 (33): 1–120. doi:10.3310/hta18330. PMC 4780997. PMID 24831822.
- ^ "Synthesis of (+)-(1R,@S,9S)-11-Methyl-7,11-Diazatricyclo[7.3.1.02.7]tridecane, a (+)sparteine surrogate". Organic Syntheses. 83: 141. 2006. doi:10.15227/orgsyn.083.0141.
- ^ The Merck Index, 10th Ed. (1983) p.402, Rahway: Merck & Co.
- ^ R. F. Keeler. Handbook of Natural Toxins: Toxicology of Plant and Fungal Compounds. CRC Press. p. 43.
- ^ Banko, P. C.; Cipollini, M. L.; Breton, G. W.; Paulk, E.; Wink, M.; Izhaki, I. (2002). "Seed chemistry of Sophora chrysophylla (mamane) in relation to diet of specialist avian seed predator Loxioides bailleui (palila) in Hawaii" (PDF). Journal of Chemical Ecology. 28 (7): 1393–410. doi:10.1023/A:1016248502927. PMID 12199503. S2CID 7064787. Archived from the original (PDF) on 2012-12-21. Retrieved 2007-05-04.
External links[]
| Wikimedia Commons has media related to Cytisine. |
- Cytisine Is Cheap And Effective In Helping Smokers Quit Information on treatment of tobacco smoking
- Reinberg, Steven. "Cheap Natural Compound May Help Smokers Quit". U.S. News & World Report. Retrieved 18 December 2014.
- Nicotinic agonists
- Alkaloids found in Fabaceae
- Lactams
- Bridged heterocyclic compounds
- Teratogens
- Plant toxins
- Nitrogen heterocycles
- Quinolizidine alkaloids
- Bulgarian inventions