Dimethylethanolamine
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-(Dimethylamino)ethan-1-ol | |
| Other names
deanol, dimethylaminoethanol, dimethylaminoethanol
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| Abbreviations | DMAE, DMEA |
Beilstein Reference
|
1209235 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.221 |
| EC Number |
|
| KEGG | |
| MeSH | Deanol |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2051 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C4H11NO |
| Molar mass | 89.138 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Fishy, ammoniacal |
| Density | 890 mg mL−1 |
| Melting point | −59.00 °C; −74.20 °F; 214.15 K |
| Boiling point | 134.1 °C; 273.3 °F; 407.2 K |
| log P | −0.25 |
| Vapor pressure | 816 Pa (at 20 °C) |
| Acidity (pKa) | 9.23 (at 20 °C)[1] |
| Basicity (pKb) | 4.77 (at 20 °C) |
Refractive index (nD)
|
1.4294 |
| Pharmacology | |
ATC code
|
N06BX04 (WHO) |
| Hazards | |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H226, H302, H312, H314, H332 |
Precautionary statements
|
P280, P305+P351+P338, P310 |
| Flash point | 39 °C (102 °F; 312 K) |
| Explosive limits | 1.4–12.2% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
| Related compounds | |
Related alkanols
|
|
Related compounds
|
Diethylhydroxylamine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
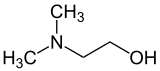
Dimethylethanolamine (DMAE or DMEA) is an organic compound with the formula (CH3)2NCH2CH2OH. It is bifunctional, containing both a tertiary amine and primary alcohol functional groups. It is a colorless viscous liquid. It is used in skin care products for improving skin tone and also taken orally as a nootropic. It is prepared by the ethoxylation of dimethylamine.[2]
Industrial uses[]
It is a precursor to other chemicals, such as the nitrogen mustard 2-dimethylaminoethyl chloride.[3] The acrylate ester is used as a flocculating agent.
Related compounds are used in gas purification, e.g. removal of hydrogen sulfide from sour gas streams.
Nutraceutical uses[]
The bitartrate salt of DMAE, i.e. 2-dimethylaminoethanol (+)-bitartrate, is sold as a dietary supplement.[4] It is a white powder providing 37% DMAE.[5]
See also[]
- Choline
- Diphenhydramine
- Doxylamine
- Ethanolamine
- Meclofenoxate
- Orphenadrine
References[]
- ^ Littel, RJ; Bos, M; Knoop, GJ (1990). "Dissociation constants of some alkanolamines at 293, 303, 318, and 333 K" (PDF). Journal of Chemical and Engineering Data. 35 (3): 276–77. doi:10.1021/je00061a014. INIST:19352048.
- ^ Matthias Frauenkron, Johann-Peter Melder, Günther Ruider, Roland Rossbacher, Hartmut Höke (2002). "Ethanolamines and Propanolamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_001. ISBN 978-3527306732.CS1 maint: uses authors parameter (link)
- ^ Ashford's Dictionary of Industrial Chemicals, 3rd edition, 2011, ISBN 978-0-9522674-3-0, p. 3294.
- ^ Karen E. Haneke & Scott Masten, 2002, "Dimethylethanolamine (DMAE) [108-01-0] and Selected Salts and Esters: Review of Toxicological Literature (Update)," Report on National Institute of Environmental Health Sciences Contract No. N01-ES-65402, November 2002, from Contractee Integrated Laboratory Systems, Research Triangle Park, North Carolina 27709, see [1], accessed 30 April 2015.
- ^ Sigma Aldrich: Safety Data Sheet: 2-Dimethylaminoethanol (+)-bitartrate
- Dimethylamino compounds
- Primary alcohols
- Cholinergics
- Nootropics
- Corrosion inhibitors
- Ethanolamines