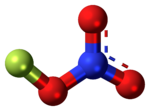
Fluorine nitrate

| |
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
Chemical formula
|
FNO3 |
| Molar mass | 81.002 g·mol−1 |
| Density | 2.217 g/L[1] |
| Melting point | −175 °C (−283.0 °F; 98.1 K) |
| Boiling point | −46 °C (−51 °F; 227 K) |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
+10.46 kJ/mol |
| Hazards | |
| Main hazards | Explosive gas |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Fluorine nitrate is an unstable derivative of nitric acid with the formula FNO
3. It is shock-sensitive.[1] Due to its instability, it is often produced from chlorine nitrate as needed[citation needed].
Synthesis and properties[]
Fluorine nitrate is formed when fluorine gas is bubbled through nitric acid or reacted with solid potassium nitrate:[2]
- F
2 + HNO
3 → FNO
3 + HF
- F
2 + KNO
3 → FNO
3 + KF
It decomposes in water to form oxygen gas, oxygen difluoride, hydrofluoric acid, and nitric acid.[1]
References[]
- ^ Jump up to: a b c Ruff, Otto; Kwasnik, Walter (1935). "The fluorination of nitric acid. The nitroxyfluoride, NO3F". Angewandte Chemie. 48: 238–240. doi:10.1002/ange.19350481604.
- ^ Yost, Don M.; Beerbower, Alan. "The Reaction of Fluorine with Nitric acid and with Solid Potassium Nitrate to Form NO3F". Communication. Cite journal requires
|journal=(help)
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
Categories:
- Inorganic compound stubs
- Nitrates
- Fluorides