PCPr
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
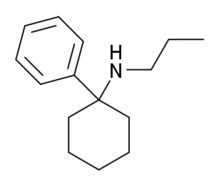
| Formula | C15H23N |
| Molar mass | 217.356 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
PCPr is an arylcyclohexylamine dissociative anesthetic drug with hallucinogenic and stimulant effects. It is around the same potency as phencyclidine, although slightly less potent than its ethyl homologue eticyclidine,[1] and has reportedly been sold as a designer drug in Germany and other European countries since the late 1990s.[2][3]
Several other related derivatives have also been encountered, with the n-propyl group of PCPr replaced by a 2-methoxyethyl, 2-ethoxyethyl or 3-methoxypropyl group to form PCMEA, PCEEA and PCMPA respectively.[4][5][6]

PCMEA, PCEEA and PCMPA
References[]
- ^ MADDOX VH, GODEFROI EF, PARCELL RF (March 1965). "The Synthesis of Phencyclidine and Other 1-Arylcyclohexylamines". Journal of Medicinal Chemistry. 8 (2): 230–5. doi:10.1021/jm00326a019. PMID 14332667.
- ^ Sauer C, Peters FT, Staack RF, Fritschi G, Maurer HH (April 2008). "Metabolism and toxicological detection of a new designer drug, N-(1-phenylcyclohexyl)propanamine, in rat urine using gas chromatography-mass spectrometry". Journal of Chromatography A. 1186 (1–2): 380–90. doi:10.1016/j.chroma.2007.11.002. PMID 18035363.
- ^ Christoph Sauer. Phencyclidine Derivatives – A new Class of Designer Drugs. Studies on the Metabolism and Toxicological Analysis. Universität des Saarlandes, 2008
- ^ Sauer C, Peters FT, Staack RF, Fritschi G, Maurer HH (March 2008). "New designer drugs N-(1-phenylcyclohexyl)-2-ethoxyethanamine (PCEEA) and N-(1-phenylcyclohexyl)-2-methoxyethanamine (PCMEA): Studies on their metabolism and toxicological detection in rat urine using gas chromatographic/mass spectrometric techniques". Journal of Mass Spectrometry. 43 (3): 305–16. Bibcode:2008JMSp...43..305S. doi:10.1002/jms.1312. PMID 17968862.
- ^ Sauer C, Peters FT, Schwaninger AE, Meyer MR, Maurer HH (February 2009). "Investigations on the cytochrome P450 (CYP) isoenzymes involved in the metabolism of the designer drugs N-(1-phenyl cyclohexyl)-2-ethoxyethanamine and N-(1-phenylcyclohexyl)-2-methoxyethanamine". Biochemical Pharmacology. 77 (3): 444–50. doi:10.1016/j.bcp.2008.10.024. PMID 19022226.
- ^ Sauer C, Peters FT, Staack RF, Fritschi G, Maurer HH (October 2008). "Metabolism and toxicological detection of the designer drug N-(1-phenylcyclohexyl)-3-methoxypropanamine (PCMPA) in rat urine using gas chromatography-mass spectrometry". Forensic Science International. 181 (1–3): 47–51. doi:10.1016/j.forsciint.2008.09.001. PMID 18922655.
Categories:
- Drugs not assigned an ATC code
- Arylcyclohexylamines
- Dissociative drugs
- Designer drugs
- NMDA receptor antagonists
- Hallucinogen stubs