Ocinaplon ATC code
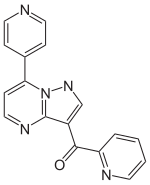
pyridin-2-yl-(7-pyridin-4-ylpyrazolo[1,5-a]pyrimidin-3-yl)methanone
CAS Number PubChem CID IUPHAR/BPS ChemSpider UNII KEGG CompTox Dashboard (EPA ) Formula C 17 H 11 N 5 O Molar mass −1 3D model (JSmol )
O=C(c1cnn2c(ccnc12)c3ccncc3)c4ncccc4
InChI=1S/C17H11N5O/c23-16(14-3-1-2-7-19-14)13-11-21-22-15(6-10-20-17(13)22)12-4-8-18-9-5-12/h1-11H
Y Key:OQJFBUOFGHPMSR-UHFFFAOYSA-N
Y
Ocinaplon is an anxiolytic drug in the pyrazolopyrimidine family of drugs. Other pyrazolopyrimidine drugs include zaleplon and indiplon .
Ocinaplon has a similar pharmacological profile to the benzodiazepine family of drugs, but with mainly anxiolytic properties and relatively little sedative or amnestic effect.[1]
Medical uses [ ] A 2019 review found tentative evidence of benefit in anxiety.[2]
Mechanism of action [ ] The mechanism of action by which ocinaplon produces its anxiolytic effects is by modulating GABAA receptors,[3] [4]
Availability [ ] Development of ocinaplon is discontinued due to liver complications that occurred in one of the Phase III subjects.[5]
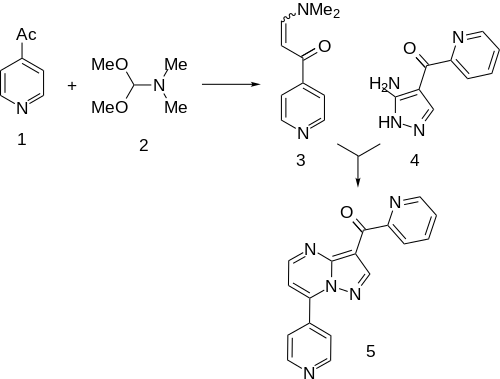
Synthesis [ ] Condensation of [8] 3 ). This is then condensed with (3-Amino-1H-pyrazol-4-yl)(2-pyridinyl)methanone (4 ) (96219-90-8).[9] [10] zaleplon in which the nitrile is replaced by a 2-acetylpyridil moiety. This affords the anxiolytic agent ocinaplon (5 ).
References [ ]
^ Lippa A, Czobor P, Stark J, Beer B, Kostakis E, Gravielle M, et al. (May 2005). "Selective anxiolysis produced by ocinaplon, a GABA(A) receptor modulator" . Proceedings of the National Academy of Sciences of the United States of America . 102 (20): 7380–5. Bibcode :2005PNAS..102.7380L . doi :10.1073/pnas.0502579102 PMC 1129138 PMID 15870187 . ^ Slee A, Nazareth I, Bondaronek P, Liu Y, Cheng Z, Freemantle N (February 2019). "Pharmacological treatments for generalised anxiety disorder: a systematic review and network meta-analysis" . Lancet . 393 (10173): 768–777. doi :10.1016/S0140-6736(18)31793-8 . PMID 30712879 . S2CID 72332967 . ^ Mirza NR, Rodgers RJ, Mathiasen LS (March 2006). "Comparative cue generalization profiles of L-838, 417, SL651498, zolpidem, CL218,872, ocinaplon, bretazenil, zopiclone, and various benzodiazepines in chlordiazepoxide and zolpidem drug discrimination". The Journal of Pharmacology and Experimental Therapeutics . 316 (3): 1291–9. doi :10.1124/jpet.105.094003 . PMID 16339395 . S2CID 21913400 . ^ Atack JR (May 2005). "The benzodiazepine binding site of GABA(A) receptors as a target for the development of novel anxiolytics". Expert Opinion on Investigational Drugs . 14 (5): 601–18. doi :10.1517/13543784.14.5.601 . PMID 15926867 . S2CID 22793644 . ^ "DOV Pharmaceutical, Inc. Places Ocinaplon Phase III Clinical Trial On Hold" . PR NewsWire . Archived from the original on 4 March 2016.^ Baumann M, Baxendale IR (October 2013). "An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles" . Beilstein Journal of Organic Chemistry . 9 : 2265–319. doi :10.3762/bjoc.9.265 . PMC 3817479 PMID 24204439 . ^ ARKIVOC 2010 (ii) 267-282 ^ LaMattina JL, Sulesk RT (1986). "A-Amino Acetals: 2,2-Diethoxy-2-(4-Pyridyl)Ethylamine". Organic Syntheses . 64 : 19. doi :10.15227/orgsyn.064.0019 . ^ U.S. Patent 4,900,836 ^ CA 1243029
Anxiolytics (N05B )
5-HT1A R agonists GABAA R PAMs
Benzodiazepines :Adinazolam Alprazolam Bromazepam Camazepam Chlordiazepoxide Clobazam Clonazepam Clorazepate Clotiazepam Cloxazolam Diazepam # Ethyl loflazepate Etizolam Fludiazepam Halazepam Ketazolam Lorazepam # Medazepam Nordazepam Oxazepam Pinazepam Prazepam ; Others: Alpidem ‡ Barbiturates (e.g., phenobarbital )Carisoprodol Carbamates (e.g., meprobamate )Chlormezanone ‡ Ethanol (alcohol) Etifoxine Imepitoin ; Herbs: Kava Skullcap Valerian Gabapentinoids (α2 δ VDCC blockers )
Gabapentin Gabapentin enacarbil Phenibut Pregabalin Antidepressants
SSRIs escitalopram )SNRIs duloxetine )SARIs trazodone )TCAs clomipramine # )TeCAs mirtazapine )MAOIs phenelzine ); Others: Agomelatine Bupropion Tianeptine Vilazodone Vortioxetine Sympatholytics (Antiadrenergics )
Alpha-1 blockers (e.g., prazosin )Alpha-2 agonists (e.g., clonidine , dexmedetomidine , guanfacine )Beta blockers (e.g., propranolol ) Others
Benzoctamine Cannabidiol Cycloserine Fabomotizole Hydroxyzine Kanna Lavender Lorpiprazole Mebicar Mepiprazole Nicotine Opipramol Oxaflozane ‡ Phenaglycodol Phenibut Picamilon Selank Tiagabine Tofisopam Validolum
# WHO-EM ‡ Withdrawn from marketClinical trials :
† Phase III § Never to phase III
GABA A receptor positive modulatorsAlcohols
Butanol Chloralodol Chlorobutanol (cloretone) Ethanol (alcohol) (alcoholic drink )Ethchlorvynol Isobutanol Isopropanol Menthol Methanol Methylpentynol Pentanol Petrichloral Propanol tert -Butanol (2M2P)tert -Pentanol (2M2B)Tribromoethanol Trichloroethanol Triclofos Trifluoroethanol Barbiturates Benzodiazepines Carbamates Flavonoids
Ampelopsin (dihydromyricetin) Apigenin Baicalein Baicalin Catechin EGC EGCG Hispidulin Luteolin Skullcap constituents (e.g., baicalin )Wogonin Imidazoles Kava constituents
Desmethoxyyangonin Kavain Methysticin Yangonin Monoureides Neuroactive steroids Nonbenzodiazepines Phenols
Fospropofol Propofol Thymol Piperidinediones Pyrazolopyridines Quinazolinones Volatiles /gases
Acetone Acetophenone Acetylglycinamide chloral hydrate Aliflurane Benzene Butane Butylene Centalun Chloral Chloral betaine Chloral hydrate Chloroform Cryofluorane Desflurane Dichloralphenazone Dichloromethane Diethyl ether Enflurane Ethyl chloride Ethylene Fluroxene Gasoline Halopropane Halothane Isoflurane Kerosine Methoxyflurane Methoxypropane Nitric oxide Nitrogen Nitrous oxide Norflurane Paraldehyde Propane Propylene Roflurane Sevoflurane Synthane Teflurane Toluene Trichloroethane (methyl chloroform) Trichloroethylene Vinyl ether Others/unsorted
3-Hydroxybutanal Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )Carbamazepine Chloralose Chlormezanone Clomethiazole DEABL Dihydroergolines (e.g., dihydroergocryptine , , dihydroergotamine , ergoloid (dihydroergotoxine) )Efavirenz Etazepine Etifoxine Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine Hopantenic acid Lanthanum Lavender oil Lignans (e.g., 4-O-methylhonokiol , honokiol , magnolol , obovatol )Loreclezole Menthyl isovalerate (validolum) Monastrol Niacin Niacinamide Org 25,435 Phenytoin Propanidid Retigabine (ezogabine) Safranal Seproxetine Stiripentol (e.g., sulfonmethane (sulfonal) , tetronal , trional )
Terpenoids (e.g., borneol )Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide , valerenic acid , )Unsorted benzodiazepine site positive modulators: α-Pinene See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators