From Wikipedia, the free encyclopedia
Anesthetic drug
Climazolam Trade names Climasol AHFS /Drugs.com International Drug Names ATCvet code Legal status
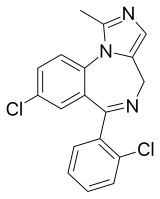
8-Chloro-6-(2-chlorophenyl)-1-methyl-4H -imidazo[1,5-a ][1,4]benzodiazepine
CAS Number PubChem CID ChemSpider UNII KEGG ChEMBL CompTox Dashboard (EPA ) Formula C 18 H 13 Cl 2 N 3 Molar mass −1 3D model (JSmol )
ClC1=CC=CC=C1C2=NCC3=CN=C(C)N3C4=C2C=C(C=C4)Cl
Climazolam [1] Ro21-3982 ) was introduced under licence as a veterinary medicine by the Swiss Pharmaceutical company Gräub under the tradename Climasol .[2] imidazo benzodiazepine derivative developed by Hoffman-LaRoche . It is similar in structure to midazolam and diclazepam and is used in veterinary medicine for anesthetizing animals.[3] [4]
References [ ]
^ US 4280957 , Walser A, Fryer RI, "Imidazodiazepines and processes therefor", issued 28 July 1981, assigned to Hoffman La Roche ^ "Climazolam" . Drugs.com .^ Ganter M, Kanngiesser M (Aug 1991). "Effect of ketamine and its combinations with xylazine and climazolam on the circulation and respiration in swine". Zentralbl Veterinarmed A (in German). 38 (7): 501–509. doi :10.1111/j.1439-0442.1991.tb01041.x . PMID 1950241 . ^ Bettschart-Wolfensberger R, Taylor PM, Sear JW, Bloomfield MR, Rentsch K, Dawling S (Oct 1996). "Physiologic effects of anesthesia induced and maintained by intravenous administration of a climazolam-ketamine combination in ponies premedicated with acepromazine and xylazine". American Journal of Veterinary Research . 57 (10): 1472–1477. PMID 8896687 .
Benzodiazepines
1,4-Benzodiazepines 1,5-Benzodiazepines 2,3-Benzodiazepines* Triazolobenzodiazepines Imidazobenzodiazepines Oxazolobenzodiazepines Thienodiazepines Thienotriazolodiazepines Thienobenzodiazepines *Pyridodiazepines Pyridotriazolodiazepines Pyrazolodiazepines Pyrrolodiazepines Tetrahydroisoquinobenzodiazepines Pyrrolobenzodiazepines *Benzodiazepine prodrugs * atypical activity profile (not GABAA receptor ligands)
GABA A receptor positive modulatorsAlcohols
Butanol Chloralodol Chlorobutanol (cloretone) Ethanol (alcohol) (alcoholic drink )Ethchlorvynol Isobutanol Isopropanol Menthol Methanol Methylpentynol Pentanol Petrichloral Propanol tert -Butanol (2M2P)tert -Pentanol (2M2B)Tribromoethanol Trichloroethanol Triclofos Trifluoroethanol Barbiturates Benzodiazepines Carbamates Flavonoids
Ampelopsin (dihydromyricetin) Apigenin Baicalein Baicalin Catechin EGC EGCG Hispidulin Luteolin Skullcap constituents (e.g., baicalin )Wogonin Imidazoles Kava constituents
Desmethoxyyangonin Kavain Methysticin Yangonin Monoureides Neuroactive steroids Nonbenzodiazepines Phenols
Fospropofol Propofol Thymol Piperidinediones Pyrazolopyridines Quinazolinones Volatiles /gases
Acetone Acetophenone Acetylglycinamide chloral hydrate Aliflurane Benzene Butane Butylene Centalun Chloral Chloral betaine Chloral hydrate Chloroform Cryofluorane Desflurane Dichloralphenazone Dichloromethane Diethyl ether Enflurane Ethyl chloride Ethylene Fluroxene Gasoline Halopropane Halothane Isoflurane Kerosine Methoxyflurane Methoxypropane Nitric oxide Nitrogen Nitrous oxide Norflurane Paraldehyde Propane Propylene Roflurane Sevoflurane Synthane Teflurane Toluene Trichloroethane (methyl chloroform) Trichloroethylene Vinyl ether Others/unsorted
3-Hydroxybutanal Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )Carbamazepine Chloralose Chlormezanone Clomethiazole DEABL Dihydroergolines (e.g., dihydroergocryptine , , dihydroergotamine , ergoloid (dihydroergotoxine) )Efavirenz Etazepine Etifoxine Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine Hopantenic acid Lanthanum Lavender oil Lignans (e.g., 4-O-methylhonokiol , honokiol , magnolol , obovatol )Loreclezole Menthyl isovalerate (validolum) Monastrol Niacin Niacinamide Org 25,435 Phenytoin Propanidid Retigabine (ezogabine) Safranal Seproxetine Stiripentol (e.g., sulfonmethane (sulfonal) , tetronal , trional )
Terpenoids (e.g., borneol )Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide , valerenic acid , )Unsorted benzodiazepine site positive modulators: α-Pinene See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators
Categories :
Benzodiazepines Chloroarenes GABAA receptor positive allosteric modulators Imidazobenzodiazepines Veterinary drugs Sedative stubs Hidden categories:
CS1 German-language sources (de) Articles with short description Short description is different from Wikidata Chemical pages without DrugBank identifier Articles without InChI source Articles containing unverified chemical infoboxes All stub articles