From Wikipedia, the free encyclopedia
The topic of this article
may not meet Wikipedia's general notability guideline .
Please help to demonstrate the notability of the topic by citing reliable secondary sources that are independent of the topic and provide significant coverage of it beyond a mere trivial mention. If notability cannot be shown, the article is likely to be merged , redirected , or deleted .Find sources: – · · · scholar · JSTOR (September 2015 ) (Learn how and when to remove this template message )
Deschloroetizolam Dependence Moderate Routes of Oral, sublingual, rectal Legal status
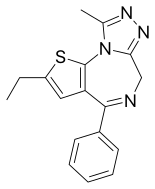
4-phenyl-2-ethyl-9-methyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazepine
CAS Number PubChem CID ChemSpider UNII Formula C 17 H 16 N 4 S Molar mass −1 3D model (JSmol )
CCC1=CC2=C(S1)N3C(=NN=C3CN=C2C4=CC=CC=C4)C
InChI=1S/C17H16N4S/c1-3-13-9-14-16(12-7-5-4-6-8-12)18-10-15-20-19-11(2)21(15)17(14)22-13/h4-9H,3,10H2,1-2H3
Key:JIOBORXCOGMHSV-UHFFFAOYSA-N
Deschloroetizolam (also known as Etizolam-2 ) is a thienotriazolodiazepine that is the dechlorinated analog of the closely related etizolam .[1] [2] [3] designer drug .[4] [5] [6]
Legal status [ ] Deschloroetizolam is classified and controlled as a hazardous substance in Sweden as of on October 15, 2015.[7]
See also [ ] Adinazolam Clonazolam Diclazepam Etizolam Flubromazepam Flubromazolam Fluetizolam Meclonazepam Metizolam Nifoxipam Pyrazolam References [ ]
^ Weber KH, Bauer A, Langbein A, Daniel H (September 1978). "Heteroaromaten mit anellierten Siebenringen, III. Umwandlung von Thienotriazolooxazepinen in Diazepine". Justus Liebigs Annalen der Chemie (in German). 1978 (8): 1257–1265. doi :10.1002/jlac.197819780806 . ^ US 3904641 , Nakanishi M, Tahara T, Araki K, Shiroki M, "Triazolothienodiazepine compounds", published 1975-09-09, assigned to Yoshitomi Pharmaceutical Industries, Ltd. ^ EP 0776892 , Hiroshi K, Syuji E, Hideaki S, Minoru M, Kenichi O, "Thienylazole compound and thienotriazolodiazepine compound", published 4 June 1997, assigned to Yoshitomi Pharmaceutical Industries, Ltd. ^ "Deschloroetizolam" . New Synthetic Drugs Database.^ Pettersson Bergstrand M, Helander A, Hansson T, Beck O (April 2017). "Detectability of designer benzodiazepines in CEDIA, EMIT II Plus, HEIA, and KIMS II immunochemical screening assays". Drug Testing and Analysis . 9 (4): 640–645. doi :10.1002/dta.2003 . PMID 27366870 . ^ Manchester KR, Maskell PD, Waters L (March 2018). "a and plasma protein binding values for benzodiazepines appearing as new psychoactive substances" (PDF) . Drug Testing and Analysis . 10 (8): 1258–1269. doi :10.1002/dta.2387 . PMID 29582576 . ^ "Nya substanser klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. 18 August 2015.
Benzodiazepines
1,4-Benzodiazepines 1,5-Benzodiazepines 2,3-Benzodiazepines* Triazolobenzodiazepines Imidazobenzodiazepines
Bretazenil Climazolam EVT-201 FG-8205 Flumazenil GL-II-73 Imidazenil 123 I-IomazenilL-655,708 Loprazolam Midazolam PWZ-029 Remimazolam Ro15-4513 Ro48-6791 Ro48-8684 Ro4938581 Sarmazenil SH-053-R-CH3-2′F Oxazolobenzodiazepines Thienodiazepines Thienotriazolodiazepines Thienobenzodiazepines *Pyridodiazepines Pyridotriazolodiazepines Pyrazolodiazepines Pyrrolodiazepines Tetrahydroisoquinobenzodiazepines Pyrrolobenzodiazepines *Benzodiazepine prodrugs * atypical activity profile (not GABAA receptor ligands)
Anxiolytics (N05B )
5-HT1A R agonists GABAA R PAMs
Benzodiazepines :Adinazolam Alprazolam Bromazepam Camazepam Chlordiazepoxide Clobazam Clonazepam Clorazepate Clotiazepam Cloxazolam Diazepam # Ethyl loflazepate Etizolam Fludiazepam Halazepam Ketazolam Lorazepam # Medazepam Nordazepam Oxazepam Pinazepam Prazepam ; Others: Alpidem ‡ Barbiturates (e.g., phenobarbital )Carisoprodol Carbamates (e.g., meprobamate )Chlormezanone ‡ Ethanol (alcohol) Etifoxine Imepitoin ; Herbs: Kava Skullcap Valerian Gabapentinoids (α2 δ VDCC blockers )
Gabapentin Gabapentin enacarbil Phenibut Pregabalin Antidepressants
SSRIs escitalopram )SNRIs duloxetine )SARIs trazodone )TCAs clomipramine # )TeCAs mirtazapine )MAOIs phenelzine ); Others: Agomelatine Bupropion Tianeptine Vilazodone Vortioxetine Sympatholytics (Antiadrenergics )
Alpha-1 blockers (e.g., prazosin )Alpha-2 agonists (e.g., clonidine , dexmedetomidine , guanfacine )Beta blockers (e.g., propranolol ) Others
Benzoctamine Cannabidiol Cycloserine Fabomotizole Hydroxyzine Kanna Lavender Lorpiprazole Mebicar Mepiprazole Nicotine Opipramol Oxaflozane ‡ Phenaglycodol Phenibut Picamilon Selank Tiagabine Tofisopam Validolum
# WHO-EM ‡ Withdrawn from marketClinical trials :
† Phase III § Never to phase III
GABA A receptor positive modulatorsAlcohols
Butanol Chloralodol Chlorobutanol (cloretone) Ethanol (alcohol) (alcoholic drink )Ethchlorvynol Isobutanol Isopropanol Menthol Methanol Methylpentynol Pentanol Petrichloral Propanol tert -Butanol (2M2P)tert -Pentanol (2M2B)Tribromoethanol Trichloroethanol Triclofos Trifluoroethanol Barbiturates Benzodiazepines Carbamates Flavonoids
Ampelopsin (dihydromyricetin) Apigenin Baicalein Baicalin Catechin EGC EGCG Hispidulin Luteolin Skullcap constituents (e.g., baicalin )Wogonin Imidazoles Kava constituents
Desmethoxyyangonin Kavain Methysticin Yangonin Monoureides Neuroactive steroids Nonbenzodiazepines Phenols
Fospropofol Propofol Thymol Piperidinediones Pyrazolopyridines Quinazolinones Volatiles /gases
Acetone Acetophenone Acetylglycinamide chloral hydrate Aliflurane Benzene Butane Butylene Centalun Chloral Chloral betaine Chloral hydrate Chloroform Cryofluorane Desflurane Dichloralphenazone Dichloromethane Diethyl ether Enflurane Ethyl chloride Ethylene Fluroxene Gasoline Halopropane Halothane Isoflurane Kerosine Methoxyflurane Methoxypropane Nitric oxide Nitrogen Nitrous oxide Norflurane Paraldehyde Propane Propylene Roflurane Sevoflurane Synthane Teflurane Toluene Trichloroethane (methyl chloroform) Trichloroethylene Vinyl ether Others/unsorted
3-Hydroxybutanal Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )Carbamazepine Chloralose Chlormezanone Clomethiazole DEABL Dihydroergolines (e.g., dihydroergocryptine , , dihydroergotamine , ergoloid (dihydroergotoxine) )Efavirenz Etazepine Etifoxine Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine Hopantenic acid Lanthanum Lavender oil Lignans (e.g., 4-O-methylhonokiol , honokiol , magnolol , obovatol )Loreclezole Menthyl isovalerate (validolum) Monastrol Niacin Niacinamide Org 25,435 Phenytoin Propanidid Retigabine (ezogabine) Safranal Seproxetine Stiripentol (e.g., sulfonmethane (sulfonal) , tetronal , trional )
Terpenoids (e.g., borneol )Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide , valerenic acid , )Unsorted benzodiazepine site positive modulators: α-Pinene See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators
PAFR
Others
Precursors
Choline ATP Phosphocholine PC LPC
See also
Receptor/signaling modulators
Categories :
Designer drugs GABAA receptor positive allosteric modulators Hypnotics Thienotriazolodiazepines Sedative stubs Hidden categories:
CS1 German-language sources (de) CS1 Swedish-language sources (sv) Articles with topics of unclear notability from September 2015 All articles with topics of unclear notability Drugs with non-standard legal status Articles without EBI source Chemical pages without DrugBank identifier Articles without KEGG source Drugs missing an ATC code Articles containing unverified chemical infoboxes All stub articles